The P-loop Motif: Decoding the ATP-Hydrolyzing Engine of NBS Domains in Disease and Drug Discovery
This comprehensive review explores the essential structural and functional role of the conserved P-loop (Walker A) motif within Nucleotide-Binding Site (NBS) domains.
The P-loop Motif: Decoding the ATP-Hydrolyzing Engine of NBS Domains in Disease and Drug Discovery
Abstract
This comprehensive review explores the essential structural and functional role of the conserved P-loop (Walker A) motif within Nucleotide-Binding Site (NBS) domains. Targeting researchers and drug developers, we first establish the evolutionary conservation and canonical architecture of the P-loop. We then detail methodological approaches for its study, including structural biology and mutational analysis, and address common challenges in experimental characterization. Finally, we compare P-loop functions across diverse protein families (e.g., ABC transporters, NLR receptors, GTPases) and validate its significance as a therapeutic target. The article synthesizes how understanding P-loop mechanics is pivotal for elucidating disease mechanisms and designing novel inhibitors.
What is the P-loop? Unveiling the Conserved Core of NBS Domain Function
The Nucleotide-Binding Site (NBS) domain is a conserved structural module central to the function of numerous proteins, including kinases, GTPases, ATPases, and NOD-like receptors (NLRs). Within the broader thesis on NBS domain conserved motifs, this whitepaper focuses on elucidating the function of the phosphate-binding loop (P-loop or Walker A motif) as the primary architectural element for coordinating nucleotide phosphates. This platform's universality lies in its ability to bind ATP or GTP, facilitating energy transduction, signal switching, and oligomerization in diverse biological pathways, making it a critical target for mechanistic research and therapeutic intervention.
Structural Anatomy of the NBS Domain
The canonical NBS domain is defined by a set of conserved sequence motifs that fold into a Rossmann-like mononucleotide-binding fold. The core motifs and their quantitative conservation metrics are summarized below.
Table 1: Conserved Motifs of the Canonical NBS Domain
| Motif Name | Consensus Sequence | Primary Function | *Conservation Frequency (%) |
|---|---|---|---|
| P-loop (Walker A) | GXXXXGK[T/S] | Binds α- and β-phosphates of NTP | >98 |
| Walker B | hhhh[D/E] (h: hydrophobic) | Coordinates Mg²⁺, activates water for hydrolysis | >95 |
| Sensor-1 | [N/T]xxx[D/E] | Stabilizes transition state, senses γ-phosphate | ~90 |
| Sensor-2 | [D/E]xx[R/K] | Couples nucleotide state to functional output | ~85 |
| A-loop | Varying | Positions adenine/guanine base | Variable |
*Frequency estimates based on curated alignments of major NBS protein families (P-loop NTPases, NLRs, kinases).
P-loop Function: Mechanism and Experimental Dissection
The P-loop (GXXXXGK[T/S]) is characterized by a glycine-rich flexible loop followed by a conserved lysine and serine/threonine. The backbone amides coordinate the β-phosphate, while the lysine side chain neutralizes the negative charge of the α- and β-phosphates. The serine/threonine often interacts with the nucleotide's ribose ring.
Key Experimental Protocol: Site-Directed Mutagenesis & ATPase Assay
Objective: To determine the functional contribution of the conserved lysine (K) in the P-loop to nucleotide binding and hydrolysis.
Detailed Methodology:
- Cloning & Mutagenesis: Clone the gene encoding the target NBS protein into an appropriate expression vector (e.g., pET-28a). Design primers to mutate the critical lysine (e.g., K→A or K→R). Perform PCR-based site-directed mutagenesis using a high-fidelity polymerase (e.g., Q5).
- Protein Expression & Purification: Transform wild-type (WT) and mutant plasmids into E. coli BL21(DE3). Induce expression with 0.5 mM IPTG at 18°C for 16 hours. Lyse cells and purify proteins via affinity chromatography (e.g., Ni-NTA for His-tagged proteins), followed by size-exclusion chromatography.
- Nucleotide Binding Assay (Fluorescence Polarization): Incubate 50 nM purified protein with a titration series (0-200 µM) of fluorescently labeled ATP/GTP analogue (e.g., BODIPY-FL-ATP) in assay buffer (20 mM Tris-HCl pH 7.5, 150 mM NaCl, 5 mM MgCl₂). Measure fluorescence polarization (FP) after 30 min equilibration. Fit data to a one-site binding model to calculate dissociation constant (Kd).
- Hydrolysis Activity Assay (Malachite Green): For ATPases, prepare reaction mix (20 mM Tris-HCl pH 7.5, 5 mM MgCl₂, 1 mM DTT, 1 mM ATP). Start reaction by adding 100 nM purified protein. At time points (0, 5, 15, 30, 60 min), quench with 0.5 M EDTA. Add malachite green reagent, incubate for 30 min, and measure A₆₂₀. Calculate liberated phosphate using a KH₂PO₄ standard curve to determine hydrolysis rate.
Table 2: Functional Impact of P-loop Lysine Mutation
| Protein Construct | Kd for ATP (µM)* | ATP Hydrolysis Rate (min⁻¹) | Relative Activity (%) |
|---|---|---|---|
| Wild-Type (WT) | 12.5 ± 1.8 | 4.2 ± 0.3 | 100 |
| K→A (Neutral) Mutant | >500 (No saturation) | 0.05 ± 0.01 | 1.2 |
| K→R (Positive) Mutant | 45.3 ± 5.1 | 1.1 ± 0.2 | 26 |
*Data representative of a typical ATPase (e.g., ABC transporter NBD). K→A abolishes binding, while K→R reduces affinity and catalysis, highlighting the precise charge and side-chain length requirement.
Diagram 1: P-loop Coordinating ATP for Hydrolysis
NBS Domain in Signaling Pathways: NLRs as a Case Study
In NLR immune receptors, the NBS domain mediates ATP-dependent oligomerization to form inflammasomes or signalosomes.
NLRP3 Inflammasome Activation Pathway
Diagram 2: NLRP3 Inflammasome Assembly via NBS
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for NBS Domain Research
| Reagent/Material | Supplier Examples | Function in NBS Research |
|---|---|---|
| Non-hydrolyzable NTP Analogs (AMP-PNP, GMP-PNP) | Jena Bioscience, Sigma-Aldrich | Traps NBS proteins in active, nucleotide-bound state for structural studies (e.g., X-ray crystallography). |
| Fluorescent NTPs (BODIPY-FL-ATP, Mant-ATP) | Thermo Fisher, Cytoskeleton Inc. | Enables real-time measurement of nucleotide binding affinity (Kd) and kinetics via FP or FRET. |
| Anti-NBS Domain Antibodies (e.g., anti-Walker A) | Abcam, Cell Signaling Technology | Detects NBS protein expression, cellular localization, and oligomerization state in Western blot/IF. |
| Malachite Green Phosphate Assay Kit | Sigma-Aldrich, Abcam | Colorimetric quantification of inorganic phosphate released in ATPase/GTPase activity assays. |
| Site-Directed Mutagenesis Kits (Q5, QuikChange) | NEB, Agilent | Generates point mutations in P-loop and other conserved motifs to probe structure-function relationships. |
| Gel Filtration/SEC Columns (Superdex 75/200) | Cytiva | Analyzes nucleotide-induced oligomerization state (monomer vs. multimer) of NBS proteins. |
| Thermal Shift Dye (SYPRO Orange) | Thermo Fisher | Monitors thermal stability shift (ΔTm) upon nucleotide binding in high-throughput screens for ligands. |
Advanced Experimental Protocol: Crystallography of an NBS-Nucleotide Complex
Objective: To obtain a high-resolution structure of an NBS domain with bound ATP analog.
Detailed Methodology:
- Protein Preparation: Express and purify NBS protein as in Section 3.1. Perform buffer exchange into crystallization buffer (e.g., 20 mM HEPES pH 7.5, 150 mM NaCl, 2 mM MgCl₂).
- Complex Formation: Incubate protein at 1.5 mM concentration with 5 mM AMP-PNP and 5 mM MgCl₂ on ice for 1 hour.
- Crystallization: Screen complex using commercial sparse-matrix screens (e.g., Hampton Research Crystal Screen) via sitting-drop vapor diffusion at 20°C. Mix 0.2 µL protein complex with 0.2 µL reservoir solution.
- Optimization: Optimize initial hits by varying pH (5.5-8.5), precipitant concentration, and adding additives (e.g., 0.1-2% benzamidine HCl).
- Data Collection & Analysis: Cryo-protect crystals in reservoir solution supplemented with 25% glycerol. Flash-cool in liquid N₂. Collect X-ray diffraction data at a synchrotron beamline. Solve structure by molecular replacement using a homologous NBS domain as a search model.
The NBS domain, through its conserved P-loop motif, provides a universal and evolutionarily optimized platform for nucleotide binding and energy coupling. Its precise mechanistic understanding, enabled by the integrative experimental approaches outlined herein, is foundational for rational drug design targeting NBS-containing proteins in cancer, autoimmune disorders, and infectious diseases. This work directly contributes to the overarching thesis by quantitatively defining the non-negotiable functional parameters of the P-loop.
This whitepaper provides an in-depth technical examination of the P-loop, also known as the Walker A motif, a critical nucleotide-binding sequence signature. This analysis is framed within a broader thesis on the functional characterization of conserved motifs within the Nucleotide-Binding Site (NBS) domain superfamily, which includes AAA+ ATPases, GTPases, kinases, and helicases. Understanding the precise sequence-structure-function relationship of the P-loop is paramount for elucidating fundamental biological processes and for targeted drug development in diseases driven by aberrant NBS domain activity.
Sequence Signature and Consensus
The P-loop is characterized by a highly conserved pattern of hydrophilic and small amino acids, facilitating phosphate backbone interaction. The canonical sequence is GxxxxGK[T/S], where 'x' represents variable, often hydrophobic, residues.
Table 1: P-loop Sequence Variations Across Protein Families
| Protein Family | Canonical Sequence (Single-Letter Code) | Conserved Position Key |
|---|---|---|
| Classic Walker A | GxxxxGK[S/T] | G1, K6, S/T7 |
| Ras-like GTPases | GxxxVGKS | G1, V5, G6, K7, S8 |
| RecA/Rad51 | GxxxxGKT | Strictly T at position 7 |
| Protein Kinases | GxGxxGxV | Often lacks the canonical lysine; part of Glycine-rich loop |
| Myosin | GESGAGKT | E2, S3 contribute to Mg²⁺ coordination |
Canonical Structure and Binding Mechanism
The P-loop forms a loop-helix structure between a β-strand (often the first in the Rossmann fold) and an α-helix. The conserved glycines provide necessary backbone flexibility. The lysine (K) side chain interacts with the β- and γ-phosphates of the nucleotide, while the serine/threonine hydroxyl coordinates the essential Mg²⁺ ion. The "P-loop" name derives from its function in binding the phosphate moiety of ATP/GTP.
Diagram 1: P-loop Nucleotide Coordination
Functional Context in NBS Domain Thesis
Within the broader thesis on NBS domains, the P-loop is the primary element responsible for anchoring and orienting the phosphates of the bound nucleotide. This initial binding event is a prerequisite for the conformational changes driven by nucleotide hydrolysis, which are transmitted to distal functional domains. Mutations in the P-loop often result in complete loss of activity or pathogenic gain-of-function, highlighting its non-redundant role.
Diagram 2: P-loop Role in NBS Domain Catalytic Cycle
Key Experimental Protocols for Analysis
Site-Directed Mutagenesis of the P-loop
Objective: To assess the functional contribution of conserved residues (e.g., G1, K6, S/T7). Protocol:
- Primer Design: Design complementary oligonucleotide primers containing the desired point mutation (e.g., K6A) flanked by 15-20 bases of wild-type sequence.
- PCR Amplification: Perform high-fidelity PCR using plasmid DNA containing the wild-type NBS domain gene as a template.
- DpnI Digestion: Treat the PCR product with DpnI endonuclease (cuts methylated DNA) to digest the parental template.
- Transformation: Transform the digested product into competent E. coli cells for plasmid circularization in vivo.
- Screening & Validation: Isolate plasmid DNA from colonies and validate the mutation by Sanger sequencing.
Fluorescent Nucleotide Binding Assay (Mant-ATP/GTP)
Objective: Quantify binding affinity (Kd) of wild-type vs. P-loop mutants. Protocol:
- Protein Purification: Express and purify recombinant wild-type and mutant NBS domain proteins (e.g., via His-tag).
- Titration: In a fluorescence cuvette, place a fixed concentration of protein (e.g., 1 µM) in assay buffer (e.g., 20 mM Tris-HCl pH 7.5, 150 mM NaCl, 5 mM MgCl₂).
- Measurement: Using a fluorometer (excitation ~360 nm, emission ~440 nm), titrate in increasing concentrations of mant-labeled nucleotide (mant-ATP or mant-GTP).
- Data Analysis: Plot fluorescence intensity (corrected for dilution and background) against nucleotide concentration. Fit data to a quadratic binding equation to determine the dissociation constant (Kd).
Crystallographic Analysis of the P-loop
Objective: Determine the atomic structure of the P-loop in complex with nucleotides. Protocol:
- Crystallization: Mix purified protein (in complex with non-hydrolyzable analogue AMP-PNP or GDP-AlF₄⁻) with crystallization screens using vapor diffusion methods.
- Data Collection: Flash-freeze crystals in liquid N₂. Collect X-ray diffraction data at a synchrotron beamline.
- Structure Solution: Solve the phase problem by molecular replacement using a homologous NBS domain structure.
- Model Building & Refinement: Iteratively build and refine the atomic model to visualize P-loop-nucleotide-Mg²⁺ coordination.
Table 2: Quantitative Data from P-loop Mutagenesis Studies
| Protein / NBS Domain | P-loop Mutation | Nucleotide Binding Affinity (Kd) Wild-type | Kd Mutant | Catalytic Rate (kcat) Wild-type | kcat Mutant | Experimental Method |
|---|---|---|---|---|---|---|
| p21 Ras (GTPase) | K16A (Walker A K) | 0.1 µM (GTP) | >100 µM (GTP) | 0.03 min⁻¹ | Not Detectable | Mant-GTP Binding, Hydrolysis |
| RecA (DNA repair) | G66A | 5 µM (ATP) | ~50 µM (ATP) | 30 min⁻¹ | <1 min⁻¹ | Fluorescent ATP Analog |
| ABC Transporter | S41A (Walker A T) | 2 µM (ATP) | 10 µM (ATP) | 10 min⁻¹ | 0.5 min⁻¹ | Radioactive ATP Filter Assay |
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Research Reagents for P-loop Studies
| Reagent / Material | Function & Application in P-loop Research |
|---|---|
| Non-hydrolyzable Nucleotide Analogues (AMP-PNP, GMP-PNP) | Used for co-crystallization and trapping NBS domains in pre-hydrolysis states to study binding geometry. |
| Mant-labeled Nucleotides (mant-ATP, mant-GTP) | Fluorescent probes for real-time monitoring of nucleotide binding affinity and kinetics via fluorescence enhancement. |
| High-Fidelity DNA Polymerase (e.g., Pfu, Q5) | Essential for error-free site-directed mutagenesis of the conserved P-loop sequence. |
| Crystallization Screening Kits (e.g., from Hampton Research) | Sparse matrix screens to identify initial conditions for growing diffraction-quality crystals of NBS domains. |
| Anti-Phospho-Substrate Antibodies | For kinases, antibodies detecting phosphorylated substrates report on functional P-loop activity in cellular assays. |
| Mg²⁺ Chelators (EDTA) & Variant Salts (MgCl₂, MnCl₂) | To probe the essential role of the divalent cation coordinated by the P-loop Ser/Thr residue. |
This whitepaper explores the profound evolutionary conservation of the Nucleotide-Binding Site (NBS) domain, with a specific focus on the canonical P-loop (Phosphate-binding loop) motif. Framed within a broader thesis on NBS domain conserved motifs, we detail the structural and functional preservation of this critical ATP/GTP-binding module from bacterial ancestors to complex human proteomes. The conserved architecture serves as a testament to a fundamental molecular mechanism essential for cellular life, presenting a premier target for cross-species therapeutic intervention.
The NBS domain is a ubiquitous structural fold responsible for binding nucleotides (ATP or GTP) across all kingdoms of life. The core functional element within this domain is the P-loop, a glycine-rich sequence motif (often GxxxxGK[T/S], where x is any amino acid) that coordinates the phosphate moiety of the bound nucleotide. Its conservation from bacterial enzymes like F1-ATPase to human proteins such as Ras GTPases, kinases, and NLR family immune receptors underscores its irreplaceable role in energy transduction, signaling, and regulation.
Quantitative Analysis of P-loop Conservation
Data synthesized from recent comparative genomic and structural analyses (2023-2024) highlight the extent of conservation.
Table 1: Prevalence of the P-loop Motif Across Major Protein Families
| Organismal Group | Key Protein Families Containing P-loop | Estimated Number of P-loop NTPases (Per Proteome) | Conserved Core Sequence |
|---|---|---|---|
| Bacteria (e.g., E. coli) | ABC Transporters, GTPases (FtsZ, Era), ATP Synthase (β subunit) | 50-100 | GxxxxGKT/S |
| Archaea | Signal Recognition Particle GTPase, Rad50-like ATPases | 40-80 | GxxxxGKT/S |
| Yeast (S. cerevisiae) | G-proteins (Ras), Kinases, AAA+ ATPases (Cdc48) | 120-150 | GxxxxGKS/T |
| Humans (H. sapiens) | Ras/MAPK pathway GTPases, Protein Kinase A, NLRP3, ABCB1 (P-gp) | 350-400 | GxxxxGKS/T |
Table 2: Structural Metrics of the Conserved P-loop Fold
| Parameter | Bacterial F1-ATPase (β subunit) | Human KRas GTPase | Human NLRP3 (NACHT domain) |
|---|---|---|---|
| PDB ID | 1BMF | 6GOD | 7PZC |
| P-loop Sequence | GGGVGKT | GAGGVGKS | GYPSGSGKS |
| Nucleotide Bound | ATP | GTP | ATP |
| Binding Affinity (Kd) | ~10 µM | ~100 pM | ~20 µM (estimated) |
| Key Coordinating Residues | Lys162 (K), Thr163 (T) | Lys16 (K), Ser17 (S) | Lys127 (K), Ser128 (S) |
Detailed Experimental Protocols for P-loop Functional Analysis
Protocol: Site-Directed Mutagenesis of the P-loop Core Lysine
Objective: To assess the functional necessity of the conserved lysine residue in nucleotide binding/hydrolysis. Materials: Target gene plasmid, mutagenic primers, high-fidelity DNA polymerase, DpnI. Procedure:
- Design forward and reverse primers containing the desired point mutation (e.g., K→A).
- Perform PCR amplification of the plasmid template using the mutagenic primers.
- Digest the parental (methylated) template DNA with DpnI for 1 hour at 37°C.
- Transform the DpnI-treated DNA into competent E. coli cells.
- Screen colonies by Sanger sequencing to confirm the mutation.
Protocol: In Vitro Nucleotide Binding Assay (Fluorescence Polarization)
Objective: To quantify the impact of P-loop mutations on nucleotide affinity. Materials: Purified wild-type and mutant proteins, fluorescently-labeled nucleotide (e.g., BODIPY-FL-GTP), assay buffer (50 mM Tris-HCl pH 7.5, 100 mM NaCl, 5 mM MgCl2), fluorescence polarization plate reader. Procedure:
- Prepare a 2x serial dilution of the protein (from 50 µM to ~0.1 µM) in a 96-well plate.
- Add a constant, low concentration (e.g., 10 nM) of BODIPY-FL-GTP to each well.
- Incubate in the dark for 30 minutes at room temperature.
- Measure fluorescence polarization (mP units) for each well.
- Fit the resulting binding curve to a one-site binding model to calculate Kd.
Protocol: Structural Analysis via X-ray Crystallography
Objective: To determine the atomic-level structural perturbations caused by P-loop mutations. Materials: Crystallized protein-nucleotide complex, synchrotron beamline access. Procedure:
- Grow crystals of the target protein (wild-type or mutant) in the presence of a non-hydrolyzable nucleotide analog (e.g., AMP-PNP, GppNHp).
- Flash-cool crystals in liquid nitrogen.
- Collect X-ray diffraction data at a synchrotron source.
- Solve the structure by molecular replacement using a known NBS domain as a search model.
- Refine the model and analyze the electron density around the P-loop and coordinated nucleotide.
Visualization of Conserved Relationships and Pathways
Diagram 1: Evolutionary conservation of the P-loop motif
Diagram 2: Generalized NBS domain signaling mechanism
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for NBS/P-loop Research
| Reagent/Material | Function/Application | Example Product/Supplier |
|---|---|---|
| Non-hydrolyzable Nucleotide Analogs (AMP-PNP, GppNHp) | Trapping NBS domains in a nucleotide-bound state for structural studies. | Jena Bioscience NU-401/405 |
| Fluorescent Nucleotides (BODIPY/ Mant- labeled) | Quantitative binding kinetics and affinity measurements (FP, FRET). | Thermo Fisher Scientific BODIPY-FL-GTP |
| Anti-P-loop/ Phospho-Nucleotide Antibodies | Detection of specific nucleotide-bound states in cells (e.g., active GTPases). | NewEast Biosciences Anti-active RAS |
| NBS Domain-Focused Mutagenesis Kits | Quick introduction of point mutations in conserved motifs (K->A, S->A). | Agilent QuikChange II |
| Thermal Shift Dye (e.g., SYPRO Orange) | Assessing protein stability and ligand binding via thermal shift assays (TSA). | Sigma-Aldrich S5692 |
| Recombinant NBS Protein Panels | Pre-purified proteins from diverse families for high-throughput screening. | R&D Systems ProKinase Panel |
| Covalent P-loop Inhibitors | Tool compounds for validating the P-loop as a drug target. | MedChemExpress MRTX1133 (KRas specific) |
Implications for Drug Development
The extreme evolutionary conservation of the P-loop presents a dual challenge and opportunity. While the structural similarity complicates achieving selectivity, it also allows for the rational design of broad-spectrum antimicrobials targeting essential bacterial NBS proteins (e.g., FtsZ, DNA gyrase). Conversely, exploiting subtle differences in the electrostatic environment or dynamics between human and pathogen P-loops is a key strategy. In human diseases, oncogenic mutations in the P-loops of Ras or kinases are primary targets, with allosteric inhibitors that stabilize inactive conformations representing a promising frontier beyond direct ATP-competitors. The functional analysis protocols outlined herein are foundational for characterizing novel inhibitors.
The P-loop motif stands as a paragon of evolutionary conservation, a minimal structural solution to the universal cellular need for nucleotide binding and hydrolysis. Research framed within the thesis of NBS domain conservation not only elucidates fundamental biology spanning bacteria to humans but also directly catalyzes the discovery of novel therapeutic agents by highlighting a primordial and essential molecular target.
Key Residues and Their Chemical Roles in ATP Coordination
Framing Thesis Context: This whitepaper details the mechanistic underpinnings of ATP coordination within the nucleotide-binding site (NBS) domain, a core focus of broader research into the structure-function relationships of the conserved phosphate-binding loop (P-loop) motif. Understanding these atomic-level interactions is paramount for elucidating enzymatic mechanisms and enabling rational drug design targeting ATP-dependent processes.
Chemical Architecture of ATP Coordination
ATP coordination within the NBS domain's P-loop is orchestrated by a conserved set of residues that form specific, often bifurcated, interactions with the phosphate chain, ribose sugar, and adenine base. The P-loop, typically with a consensus sequence of GXXXXGK[T/S] (where X is any amino acid), provides the scaffold for anchoring the triphosphate moiety.
Key Residues and Their Primary Chemical Roles
- Lysine in the P-loop (e.g., K15 in PKA): Its positively charged ε-amino group forms critical ionic interactions with the negatively charged β- and γ-phosphates of ATP. It also stabilizes the transition state during phosphoryl transfer.
- Serine/Threonine in the P-loop (e.g., T/S17 in PKA): The hydroxyl group coordinates the essential Mg²⁺ ion that bridges the β- and γ-phosphates, neutralizing charge and orienting ATP for catalysis.
- Aspartate/Gluatamate in the DFG/DMG Motif (Walker B): Coordinates the second essential Mg²⁺ ion, which interacts with the α- and β-phosphates, and often acts as a catalytic base.
- "Lid" Residue (e.g., R70 in PKA, often an arginine): From a non-P-loop motif, it forms a bidentate salt bridge with the α- and γ-phosphates, sealing ATP in the binding pocket and providing transition state stabilization.
- Adenine-Binding Residues: Typically hydrophobic residues (e.g., valine, alanine) form van der Waals contacts with the adenine ring, while a backbone carbonyl often hydrogen-bonds with N6 and N1 of adenine.
Quantitative Analysis of Interactions
Table 1: Representative Bond Distances and Energies in ATP Coordination (Compiled from PDB analyses & QM/MM studies)
| Residue (Role) | Atom Pair (Residue-ATP) | Typical Distance (Å) | Interaction Type | Estimated Energy Contribution |
|---|---|---|---|---|
| P-loop Lysine | NZ - O (β-phosphate) | 2.7 - 3.1 | Ionic / H-bond | -15 to -25 kcal/mol (in cluster) |
| P-loop Ser/Thr | OG - Mg²⁺ | 2.0 - 2.2 | Coordination | - |
| Catalytic Asp/Glu | OD - Mg²⁺ | 2.0 - 2.2 | Coordination | - |
| "Lid" Arginine | NH1 - O (γ-phosphate) | 2.8 - 3.2 | Bidentate Ionic | -20 to -30 kcal/mol |
| Backbone Carbonyl | C=O - N6 (Adenine) | 2.9 - 3.2 | H-bond (backbone) | -3 to -6 kcal/mol |
| Mg²⁺ Ion (M1) | Mg²⁺ - O (β, γ-P) | 2.0 - 2.2 | Ionic Coordination | Significant charge shielding |
Experimental Protocols for Investigating ATP Coordination
Protocol: Site-Directed Mutagenesis Coupled with Kinetic Assays
Objective: To determine the functional contribution of a specific residue to ATP binding and hydrolysis. Methodology:
- Design Primers: Design oligonucleotide primers containing the desired nucleotide mutation for the target residue (e.g., Lys→Ala to remove charge).
- PCR Mutagenesis: Perform polymerase chain reaction (PCR) using a plasmid containing the wild-type kinase/NBS domain gene as a template.
- DpnI Digestion: Treat PCR product with DpnI endonuclease to digest the methylated parental DNA template.
- Transformation & Sequencing: Transform into competent E. coli, plate, and pick colonies for plasmid DNA sequencing to confirm the mutation.
- Protein Expression & Purification: Express and purify the wild-type and mutant proteins using affinity chromatography (e.g., Ni-NTA for His-tagged proteins).
- Enzymatic Kinetics:
- Perform ATPase or kinase activity assays using [γ-³²P]ATP or a coupled spectrophotometric assay (e.g., with lactate dehydrogenase/pyruvate kinase).
- Vary ATP concentration and measure initial velocity.
- Fit data to the Michaelis-Menten equation to determine Km (ATP) and kcat.
- Analysis: Compare mutant and wild-type kinetics. A increased Km suggests impaired ATP binding; a decreased kcat suggests impaired catalysis/transition state stabilization.
Protocol: X-ray Crystallography of NBS Domain-ATP Analog Complexes
Objective: To obtain atomic-resolution structure of the coordination network. Methodology:
- Protein Crystallization: Co-crystallize the purified NBS domain protein with a non-hydrolyzable ATP analog (e.g., AMP-PNP, ATPγS) and essential Mg²⁺ or Mn²⁺ ions via vapor diffusion.
- Cryoprotection & Flash-Cooling: Transfer crystal to a cryoprotectant solution (e.g., 25% glycerol) and flash-cool in liquid nitrogen.
- Data Collection: Collect X-ray diffraction data at a synchrotron source.
- Structure Solution & Refinement:
- Solve the phase problem by molecular replacement using a known homologous structure.
- Iteratively refine the model (coordinates and B-factors) and build the ligand (ATP analog, ions) into clear electron density (Fo-Fc map).
- Analysis: Measure atomic distances and angles of interactions between protein residues, metal ions, and the ATP analog using software like PyMOL or Coot.
Visualization of ATP Coordination Network
Diagram Title: ATP Coordination Network in the NBS Domain P-loop
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Research Reagent Solutions for ATP Coordination Studies
| Reagent / Material | Function / Application | Key Consideration |
|---|---|---|
| Non-hydrolyzable ATP Analogs (AMP-PNP, ATPγS) | Co-crystallization or trapping of ATP-bound states for structural studies; inhibits activity for kinetic assays. | Choose based on structural mimicry (AMP-PNP) or sulfur substitution for spectroscopy (ATPγS). |
| Radioisotope [γ-³²P]ATP | Sensitive detection of phosphate transfer in kinase/ATPase activity assays. | Requires radiation safety protocols; used in filter-binding or gel-based assays. |
| MgCl₂ / MnCl₂ Solutions | Source of essential divalent cations (Mg²⁺, Mn²⁺) for ATP coordination and catalysis. Mn²⁺ is often used for phasing in crystallography. | Concentration is critical; typically 5-20 mM excess over ATP. |
| Site-Directed Mutagenesis Kit (e.g., QuikChange) | Efficiently introduces point mutations into the gene of interest to probe residue function. | Primer design is crucial for success and specificity. |
| Fast Protein Liquid Chromatography (FPLC) System | High-resolution purification of recombinant wild-type and mutant NBS domain proteins (e.g., via size-exclusion, ion-exchange). | Essential for obtaining homogeneous, active protein for kinetics and crystallography. |
| Crystallization Screening Kits (e.g., from Hampton Research) | Sparse-matrix screens to identify initial conditions for protein/ATP analog co-crystallization. | Includes varied PEGs, salts, and buffers to sample chemical space. |
| Coupled Enzyme Assay System (PK/LDH) | Spectrophotometric continuous assay for ATPase activity, measuring ADP production via NADH oxidation. | Allows real-time kinetics; requires optimization to ensure coupling enzyme is not rate-limiting. |
This whitepaper addresses a central pillar of the broader thesis on Nucleotide-Binding Site (NBS) domain conserved motifs, focusing on the P-loop (Phosphate-binding loop, Walker A motif). The thesis posits that the P-loop is not merely a passive phosphate-binding structure but a thermodynamically optimized catalytic scaffold that actively facilitates phosphoryl transfer—a fundamental reaction in bioenergetics and signaling. This document provides an in-depth technical examination of the thermodynamic and mechanistic principles underlying this function, supported by current structural and biophysical data.
Structural and Thermodynamic Foundation of the P-loop
The canonical P-loop sequence (GxxxxGK[T/S], where 'x' is any residue) forms a flexible loop between a beta-strand and an alpha-helix. Its function is underpinned by precise thermodynamic contributions:
- Entropic Pre-organization: The glycine-rich backbone provides conformational flexibility, allowing the loop to "close" upon nucleotide binding. This reduces the entropic penalty associated with ordering the substrate and catalytic moieties, a key step in lowering the activation energy.
- Enthalpic Stabilization: The conserved lysine and serine/threonine residues form direct hydrogen bonds with the beta- and gamma-phosphates of ATP or GTP. This binding energy stabilizes the transition state of the phosphoryl transfer reaction, which often involves a pentavalent phosphorus intermediate.
- Mg²⁺ Coordination: The P-loop, along with conserved aspartate residues from Walker B or other motifs, coordinates a catalytically essential Mg²⁺ ion. This ion neutralizes the negative charge on the phosphate groups, making the gamma-phosphorus more electrophilic and susceptible to nucleophilic attack.
Table 1: Thermodynamic and Kinetic Parameters of P-loop Mediated Phosphoryl Transfer
| Parameter | Typical Range in P-loop NTPases | Significance |
|---|---|---|
| Kd for NTP | 0.1 - 10 µM | High-affinity binding driven by P-loop interactions. |
| ΔG of NTP Binding | -40 to -50 kJ/mol | Favorable free energy change enabling stable complex formation. |
| Catalytic Rate (kcat) | 1 - 1000 s⁻¹ | Direct measure of phosphoryl transfer efficiency. |
| Mg²⁺ Concentration for Max Activity | 1 - 5 mM | Highlights essential role of divalent cation coordination. |
| Activation Energy (Ea) Reduction | ~20-30 kJ/mol | Quantifies the P-loop's role in transition state stabilization. |
Detailed Experimental Protocol: Isothermal Titration Calorimetry (ITC) for Binding Thermodynamics
Objective: To quantify the enthalpy (ΔH), entropy (ΔS), and binding constant (Ka) of ATP binding to a purified P-loop containing protein (e.g., a kinase or GTPase).
Methodology:
- Sample Preparation:
- Purify recombinant P-loop protein to homogeneity (>95% purity) via affinity and size-exclusion chromatography.
- Dialyze protein and ATP stock into identical buffer (e.g., 20 mM HEPES pH 7.5, 150 mM NaCl, 5 mM MgCl₂). Centrifuge to remove particulates.
- Determine precise protein concentration using absorbance at 280 nm.
ITC Experiment:
- Load the sample cell (1.4 mL) with protein solution (10-50 µM).
- Fill the syringe with ATP solution (10x the protein concentration).
- Set reference power to 10-15 µcal/sec and cell temperature to 25°C.
- Program injections: 1 initial 0.5 µL injection (discarded), followed by 19-28 injections of 2-10 µL each, spaced 180-240 seconds apart.
- Perform a control titration of ATP into buffer to subtract dilution heat.
Data Analysis:
- Integrate raw heat peaks and subtract control titration data.
- Fit the binding isotherm to a single-site binding model using the instrument's software.
- The fit yields n (stoichiometry), Ka (association constant), and ΔH (enthalpy change).
- Calculate ΔG = -RT lnKa and ΔS = (ΔH - ΔG)/T.
Key Signaling Pathways Involving P-loop NTPases
P-loop NTPases (GTPases and kinases) are nodal points in cellular signaling. The diagram below illustrates a generalized GTPase signaling cycle, central to the thesis on motif function.
Diagram Title: GTPase Signaling Cycle Featuring P-loop
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for P-loop Function Research
| Reagent / Material | Function in Research | Key Consideration |
|---|---|---|
| Non-hydrolyzable NTP Analogs (e.g., AMP-PNP, GTPγS) | Traps protein in pre- or post-transfer state for structural studies (X-ray, Cryo-EM). | Mimics natural substrate but prevents reaction turnover. |
| Fluorescent NTPs (e.g., Mant-ATP, BODIPY-GTP) | Monitors binding kinetics and conformational changes via FRET or fluorescence polarization. | Fluorophore should minimally perturb P-loop binding. |
| Transition State Analogs (e.g., AlF3, BeF3⁻, VO4³⁻) | Mimics the pentavalent transition state of hydrolysis; stabilizes protein-GDP-analog complexes. | Essential for crystallizing catalytic intermediates. |
| High-Purity Mg²⁺/Mn²⁺ Salts | Provides essential catalytic cation. Mn²⁺ is often used for EPR spectroscopy. | Contaminants (e.g., Ca²⁺) can inhibit activity; use ultrapure grade. |
| Site-Directed Mutagenesis Kits | Creates mutations in P-loop residues (e.g., K→A, S→A) to dissect functional contributions. | Critical for establishing structure-function relationships. |
| ITC or SPR Instrumentation & Consumables | Directly measures binding thermodynamics (ITC) or kinetics (SPR) of NTP binding. | Requires highly pure, monodisperse protein samples. |
Experimental Workflow for Mechanistic Analysis
The following diagram outlines a integrated workflow to dissect P-loop function, as employed in contemporary research.
Diagram Title: Integrated P-loop Analysis Workflow
The P-loop operates on a core thermodynamic principle: it pre-organizes the active site to pay the entropic cost of catalysis upfront and uses precise, enthalpically favorable interactions to stabilize the high-energy transition state of phosphoryl transfer. This principle, validated by the experimental data and protocols detailed herein, is a unifying feature across the vast superfamily of P-loop NBS domain proteins. For the broader thesis, this underscores the P-loop's evolution as a versatile and optimal solution for controlling the energy currency of the cell, making it a critical target for rational drug design in oncology, infectious disease, and beyond.
How to Study the P-loop: Techniques from Structural Biology to Mutagenesis
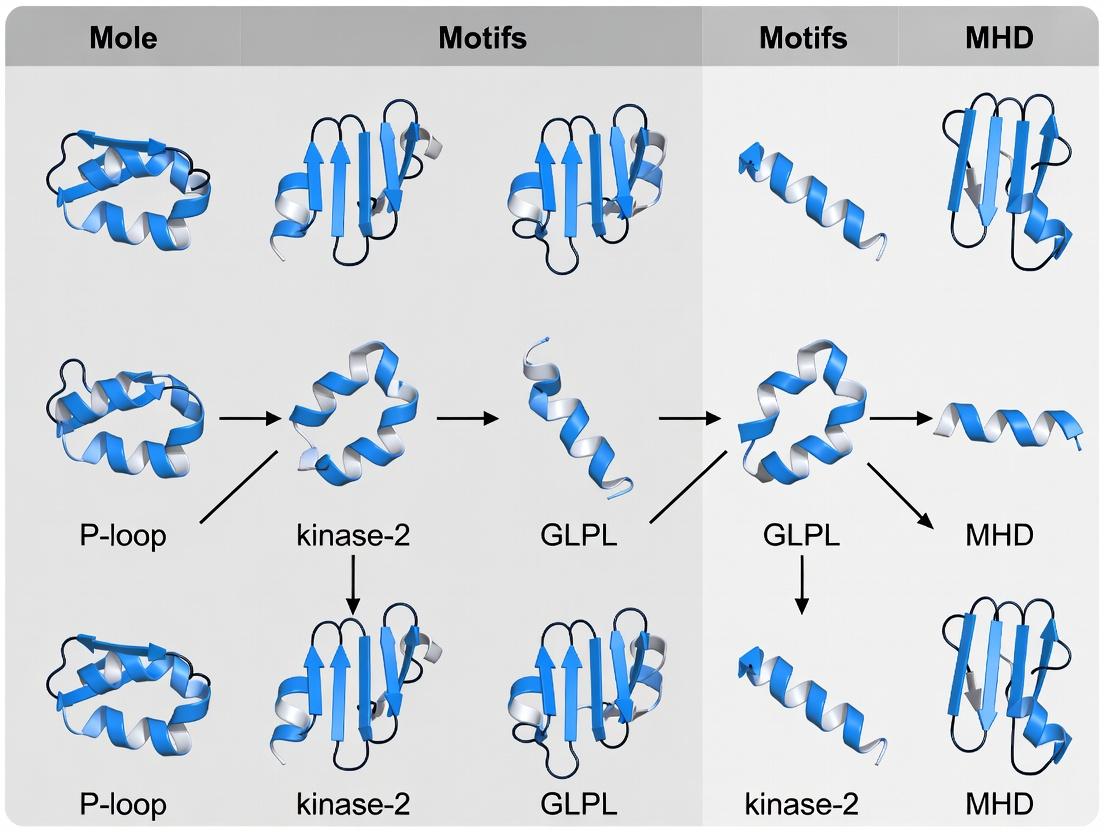
Nucleotide-binding site (NBS) domains are a hallmark of signal transduction ATPases, central to innate immune receptors, apoptosis regulators (APAF-1), and plant disease resistance proteins. The broader thesis of P-loop function research posits that conserved motifs within the NBS—notably the P-loop (Walker A), Walker B, Sensor 1, and RNBS motifs—orchestrate nucleotide-dependent conformational switches that regulate protein activity, oligomerization, and downstream signaling. Structural elucidation via X-ray crystallography and cryo-electron microscopy (cryo-EM) is indispensable for visualizing these mechanistic details at atomic resolution, enabling the rational design of therapeutics that modulate immune and cell death pathways.
Table 1: Comparison of Structural Insights from X-ray Crystallography and Cryo-EM for Key NBS Domain Proteins
| Protein (PDB/EMDB ID) | Method | Resolution (Å) | Key NBS Motifs Resolved | Oligomeric State Observed | Nucleotide State | Key Insight for P-loop Function |
|---|---|---|---|---|---|---|
| NLRC4 (6B5B) | X-ray | 3.3 | P-loop, Walker B, HD1 | Inactive monomer | None | P-loop lysine coordinates with α-phosphate in ADP-bound state. |
| APAF-1 (6RFD) | Cryo-EM | 3.8 | P-loop, Walker B, Sensor 1 | Heptameric apoptosome | dATP/ATP bound | P-loop engages nucleotide, enabling ring assembly. |
| Mouse NLRP3 (7PZC) | Cryo-EM | 3.0 | P-loop, Walker B, WHD | Dodecameric disk | ADP bound | P-loop interacts with ADP; conformation inhibits activity. |
| ZAR1 (6J5W) | X-ray | 3.6 | P-loop, RNBS-A, MHD | Pentameric resistosome | ADP bound | P-loop binds ADP; MHD motif acts as nucleotide sensor. |
| hNLRP1 (7LD1) | Cryo-EM | 3.3 | P-loop, Walker B, FISNA | Dodecameric disk | ATP bound | ATP binding stabilizes active oligomer via P-loop. |
Table 2: Conserved NBS Motifs and Their Functional Roles
| Motif Name | Consensus Sequence | Structural Role | Functional Implication for P-loop Thesis |
|---|---|---|---|
| Walker A (P-loop) | GXXXXGK[T/S] | Binds α- and β-phosphates of nucleotide. | Initiates nucleotide binding; GK[S/T] interacts with Mg²⁺, essential for hydrolysis. |
| Walker B | hhhh[D/E] | Coordinates Mg²⁺; glutamate activates water for hydrolysis. | Couples nucleotide state to conformational change in Sensor 1. |
| Sensor 1 | [T/S]xxxN | Hydrogen-bonds to Walker B via conserved Asn. | Transduces hydrolysis signal to the rest of the domain. |
| RNBS-A | [F/Y]xGxP | Forms hydrophobic core; stabilizes NBS fold. | Mutations disrupt NBS stability, ablating P-loop function. |
| MHD / Sensor 2 | MHD | Binds ribose and γ-phosphate; senses nucleotide state. | Key discriminator between ATP/ADP, controlling activation switch. |
Experimental Protocols for Structural Elucidation
Protocol 3.1: X-ray Crystallography of an NBS Domain Protein
Objective: Determine high-resolution structure of an NLR NBS domain in complex with ADP.
- Protein Expression & Purification:
- Express His-tagged NBS protein in E. coli BL21(DE3). Purify via Ni-NTA affinity, followed by size-exclusion chromatography (SEC) in buffer: 20 mM HEPES pH 7.5, 150 mM NaCl, 5 mM MgCl₂, 1 mM TCEP.
- Crystallization:
- Mix protein (10 mg/mL + 5 mM ADP) with reservoir solution at 1:1 ratio using sitting-drop vapor diffusion.
- Condition: 0.1 M HEPES pH 7.0, 25% (w/v) PEG 3350, 0.2 M ammonium acetate. Incubate at 18°C.
- Data Collection & Processing:
- Flash-cool crystal in liquid N₂ with 20% ethylene glycol as cryoprotectant.
- Collect 360° of data at a synchrotron beamline (λ = 0.9786 Å). Process with XDS or DIALS. (Typical stats: Resolution 3.0 Å, Rmerge < 10%, completeness > 95%).
- Structure Solution & Refinement:
- Solve via molecular replacement (Phaser) using a homologous NBS structure (e.g., NLRC4).
- Iteratively refine with Phenix.refine and manually build in Coot. Validate with MolProbity.
Protocol 3.2: Cryo-EM Analysis of an Activated NBS Oligomer (e.g., Apoptosome)
Objective: Determine structure of a large, nucleotide-activated NBS assembly.
- Sample Preparation:
- Assemble APAF-1 apoptosome by incubating purified APAF-1 with cytochrome c and dATP for 1 hr at 30°C.
- Apply 3 μL of sample (0.5 mg/mL) to a glow-discharged Quantifoil R1.2/1.3 Au grid. Blot (3s, blot force -5) and plunge-freeze in liquid ethane using Vitrobot (100% humidity, 4°C).
- Data Acquisition:
- Collect movies (40 frames, total dose 50 e⁻/Ų) on a 300 kV Titan Krios with a K3 direct electron detector. Use SerialEM for automated collection (1.05 Å/pixel). Target 5,000-10,000 micrographs.
- Image Processing:
- Motion-correct with MotionCor2, CTF-estimate with CTFFIND-4.2.
- Pick particles with cryoSPARC blob picker, extract, and perform 2D classification.
- Generate ab initio models, followed by heterogeneous refinement to isolate intact oligomers.
- Run non-uniform refinement and local refinement to achieve final map (3.5-4.0 Å resolution).
- Model Building & Refinement:
- Fit an available X-ray structure into the EM density using ChimeraX.
- Manually adjust in Coot and perform real-space refinement in Phenix.
Visualization of Signaling Pathways and Workflows
Diagram Title: NBS Domain Activation via Nucleotide Switch
Diagram Title: X-ray vs Cryo-EM Structural Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for NBS Domain Structural Studies
| Item | Function in NBS Research | Example Product / Note |
|---|---|---|
| Expression Vector | High-yield protein production for crystallization. | pET series (Novagen) with N-terminal His-tag. |
| Affinity Resin | Initial purification of tagged NBS protein. | Ni Sepharose High Performance (Cytiva). |
| Size-Exclusion Column | Polishing step to obtain monodisperse, oligomeric samples. | Superdex 200 Increase 10/300 GL (Cytiva). |
| Nucleotides | Essential cofactors for NBS domain stability and activity. | ATP (A2383), ADP (A2754), dATP (D6500) from Sigma. Use with MgCl₂. |
| Crystallization Screen Kits | Initial search for crystallization conditions. | MORPHEUS (Molecular Dimensions) for membrane proteins/soluble complexes. |
| Cryo-EM Grids | Support film for vitrified sample. | Quantifoil R1.2/1.3 300 mesh Au or UltrauFoil. |
| Vitrification System | Rapid freezing to preserve hydrated state. | Thermo Fisher Vitrobot Mark IV. |
| Image Processing Software | Transforming micrographs into 3D density maps. | cryoSPARC Live (Structura Biotechnology), Relion. |
| Model Refinement Suite | Building and validating atomic coordinates. | Phenix (phenix.refine), Coot for manual building. |
The nucleotide-binding site (NBS) domain, characterized by conserved phosphate-binding (P-loop or Walker A) and hydrolysis (Walker B) motifs, is a critical functional module in ATPases and kinases. Research into its precise molecular mechanism—how it coordinates Mg²⁺-ATP, stabilizes the transition state, and couples hydrolysis to conformational changes—relies fundamentally on quantitative biochemical assays. This technical guide details the core methodologies for measuring ATP binding, hydrolysis, and kinetics, providing the experimental framework essential for probing P-loop function in structural biology, enzymology, and targeted drug development.
Core Quantitative Assays: Principles and Data
The following assays provide complementary data on different stages of the ATPase cycle.
Table 1: Core Assays for ATP Interaction Analysis
| Assay Type | What it Measures | Key Readout | Typical Application in P-loop Studies |
|---|---|---|---|
| Isothermal Titration Calorimetry (ITC) | Thermodynamics of binding (KD, ΔH, ΔS, n). | Heat change per injection. | Direct measurement of ATP affinity to wild-type vs. mutant P-loop. |
| Surface Plasmon Resonance (SPR) | Association (kon) and dissociation (koff) rates, affinity (KD). | Resonance units (RU) vs. time. | Real-time analysis of ATP analog binding kinetics. |
| Enzyme-Coupled NADH Assay | Steady-state hydrolysis rate (v0). | Decrease in A340 from NADH oxidation. | Determining Michaelis-Menten constants (KM, kcat). |
| Malachite Green Phosphate Assay | End-point inorganic phosphate (Pi) release. | Increase in A620/A650. | High-throughput screening of hydrolysis inhibitors. |
| Radioactive [γ-32P]ATP Assay | Real-time Pi release or ATP-ADP exchange. | Radiolabeled Pi or ADP detected. | Single-turnover kinetics and mechanistic studies. |
Table 2: Representative Kinetic Parameters from P-loop Mutant Studies
| Protein (Mutation) | KD for ATP (μM) | kcat (s⁻¹) | KM for ATP (μM) | Catalytic Efficiency (kcat/KM, M⁻¹s⁻¹) |
|---|---|---|---|---|
| Wild-type NBS Domain | 15.2 ± 2.1 | 120 ± 10 | 25 ± 3 | 4.8 x 10⁶ |
| P-loop GXXGXGK → GXXGXGA | 210 ± 35 | 1.5 ± 0.3 | 300 ± 45 | 5.0 x 10³ |
| Walker B D → N | 18.5 ± 3.0 | 0.8 ± 0.1 | 28 ± 4 | 2.9 x 10⁴ |
Detailed Experimental Protocols
Protocol 1: Enzyme-Coupled Assay for Steady-State Kinetics Objective: Determine kcat and KM for ATP hydrolysis.
- Reaction Mix: Prepare 100 µL reactions containing: 50 mM Tris-HCl (pH 7.5), 10 mM MgCl₂, 1 mM DTT, 0.2 mM NADH, 1 mM phospho(enol)pyruvate, 5 units/mL pyruvate kinase, 5 units/mL lactate dehydrogenase, and varying [ATP] (e.g., 0.1-5 x estimated KM).
- Initiation: Pre-incubate mix + enzyme (5-100 nM) for 2 min at assay temperature (e.g., 30°C). Initiate reaction by adding ATP.
- Data Acquisition: Monitor A340 continuously for 5-10 min in a plate reader or spectrophotometer.
- Analysis: Calculate initial velocity (v0) from the linear slope (εNADH = 6220 M⁻¹cm⁻¹). Fit v0 vs. [ATP] to the Michaelis-Menten equation: v0 = (Vmax[S])/(KM+[S]).
Protocol 2: ITC for Direct Binding Affinity Objective: Measure the thermodynamic profile of ATP binding to an NBS domain protein.
- Sample Prep: Dialyze protein (>50 µM) and ATP ligand into identical buffer (e.g., 20 mM HEPES, pH 7.5, 150 mM NaCl, 5 mM MgCl₂). Degas samples.
- Titration: Load the cell with protein. Fill syringe with ATP (10-20x stock concentration). Set instrument temperature (25°C).
- Parameters: Perform 15-20 injections (2-4 µL each) with 150-180 sec spacing. Use a reference cell with water.
- Analysis: Integrate heat peaks per injection. Subtract control (ligand into buffer). Fit binding isotherm to a single-site model to derive n, KD, ΔH, and ΔS.
Signaling and Experimental Pathways
Title: ATP Hydrolysis Cycle Catalyzed by NBS Domain P-loop.
Title: Experimental Workflow for P-loop Function Analysis.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for ATPase Assays
| Reagent / Material | Function & Role in Assay |
|---|---|
| High-Purity ATP (e.g., Na₂ATP) | Primary substrate. Requires titration for exact concentration and Mg²⁺盐 to form MgATP complex. |
| MgCl₂ / MgSO₄ | Essential cofactor. Stabilizes ATP and is often coordinated by P-loop residues. |
| ITC Instrument (e.g., Malvern PEAQ-ITC) | Directly measures heat from binding interactions. Gold standard for label-free KD. |
| Enzyme Coupling System (PK/LDH, NADH, PEP) | Couples ADP production to oxidation of NADH, enabling continuous spectrophotometric readout. |
| Malachite Green Reagent Kit | Colorimetric detection of free inorganic phosphate (Pi) for end-point, high-throughput assays. |
| [γ-32P]ATP | Radioactive tracer for highly sensitive, single-turnover kinetic studies of Pi release. |
| SPR Chip (e.g., NTA for His-tagged proteins) | Immobilizes protein for real-time analysis of binding kinetics (kon, koff). |
| Non-hydrolyzable ATP Analogs (AMP-PNP, ATPγS) | Used in structural and binding studies to trap the pre-hydrolysis state. |
| P-loop/Walker Motif Mutant Proteins | Key reagents generated via site-directed mutagenesis to dissect motif-specific contributions. |
1. Introduction This whitepaper details a core methodological and analytical framework for probing the function of the Phosphate-binding loop (P-loop), a highly conserved motif within Nucleotide-Binding Site (NBS) domains. As part of a broader thesis on NBS domain conserved motifs, functional knockout via site-directed mutagenesis (SDM) of P-loop residues serves as a definitive approach to elucidate their non-redundant roles in nucleotide binding, hydrolysis, and subsequent signal transduction in ATPases and GTPases, including kinases, G-proteins, and NLR immune receptors.
2. Target Residue Selection & Rationale The canonical P-loop sequence is commonly denoted as G-X-X-X-X-G-K-T/S, where 'X' represents variable residues. Key conserved residues are prime targets for mutagenesis.
Table 1: Conserved P-loop Residues and Rationale for Mutagenesis
| Residue Position | Conserved Role | Common Substitution(s) | Predicted Functional Impact |
|---|---|---|---|
| First Glycine | Main chain flexibility, nucleotide positioning. | G→A (Alanine) | Steric hindrance, reduced loop flexibility, impaired nucleotide docking. |
| Lysine | Stabilizes β- and γ-phosphates; catalytic mantle. | K→A (Alanine), K→R (Arginine) | Loss of charge interaction; R tests charge preservation with altered geometry. |
| Threonine/Serine | Coordinates Mg²⁺ ion via main chain carbonyl and side chain. | T→A (Alanine), T→V (Valine) | Loss of Mg²⁺ coordination, crippling catalytic activity. |
| Second Glycine | Allows close approach of Walker A and B motifs. | G→A (Alanine) | Disruption of inter-motif geometry, affecting hydrolysis. |
3. Detailed Experimental Protocol: SDM & Functional Assay
3.1. Primer Design for Site-Directed Mutagenesis (PCR-based)
- Method: High-fidelity, non-strand displacing polymerase (e.g., Phusion, Q5) is used in a PCR reaction with plasmid DNA as template.
- Primer Design Rules: Primers are typically 25-45 bases, complementary to opposite strands of the plasmid. The desired mutation is placed centrally. Both primers must contain the mutation. A melting temperature (Tm) ≥78°C is recommended for the primer-template complex.
- Example Primer Pair for K→A Mutation:
- Forward: 5’-CCT GGA GTA G C A TTA GCT GGT ACC GAC-3’ (Mutated base in bold)
- Reverse: 5’-GTC GGT ACC AGC TAA TG C T ACT CCA GG-3’
- PCR Protocol:
- Mix: 10-50 ng plasmid template, 0.5 µM each primer, 200 µM dNTPs, 1X HF buffer, 1 unit polymerase in 50 µL.
- Cycle: 98°C for 30 sec; [98°C for 10 sec, Tm+3°C for 20 sec, 72°C for 2-4 min/kb] x 25 cycles; 72°C for 5 min.
- Digest parental template: Add 1 µL DpnI restriction enzyme directly to PCR product, incubate at 37°C for 1-4 hours. DpnI cleaves methylated (template) DNA.
- Transform 5 µL of DpnI-treated DNA into competent E. coli, plate on selective media.
- Screen colonies by Sanger sequencing of the entire NBS domain region.
3.2. Functional Assay: In vitro Nucleotide Binding & Hydrolysis
- Protein Purification: Express wild-type (WT) and mutant proteins (e.g., with His₆-tag) and purify via affinity chromatography.
- Radiolabeled Nucleotide Binding Filter Assay:
- Incubate 1-10 pmol of purified protein in binding buffer (e.g., 50 mM Tris-HCl pH 7.5, 50 mM NaCl, 5 mM MgCl₂) with increasing concentrations of [α-³²P]ATP/GTP (e.g., 0.1-10 µM) for 15 min at 25°C.
- Filter the reaction through a nitrocellulose membrane (binds protein-nucleotide complexes).
- Wash membrane 3x with ice-cold binding buffer.
- Measure membrane-bound radioactivity by scintillation counting.
- Analyze data using nonlinear regression to determine dissociation constant (Kd).
- Coupled Enzymatic Hydrolysis Assay (ATPase/GTPase):
- Use a coupled system where ADP/GDP production is linked to NADH oxidation, monitored at 340 nm.
- Reaction mix: 50 mM Tris-HCl pH 7.5, 100 mM KCl, 5 mM MgCl₂, 2 mM phosphoenolpyruvate, 0.2 mM NADH, 10 U/ml pyruvate kinase, 10 U/ml lactate dehydrogenase, 1-5 µM protein, 1-5 mM ATP/GTP.
- Initiate reaction by adding nucleotide, monitor A₃₄₀ decrease for 10-30 min at 25-30°C.
- Calculate hydrolysis rate (µM NADH oxidized/min = µM Pi released/min).
Table 2: Example Functional Data from P-loop Mutant Analysis
| Protein Variant | Nucleotide Kd (µM) | % Binding vs. WT | Hydrolysis Rate (min⁻¹) | % Activity vs. WT |
|---|---|---|---|---|
| WT (Control) | 1.2 ± 0.2 | 100% | 25.0 ± 2.1 | 100% |
| G→A (1st Gly) | 15.5 ± 3.1 | ~8% | 0.5 ± 0.2 | 2% |
| K→A | 5.8 ± 1.2 | ~21% | 1.2 ± 0.3 | 5% |
| T→A | 2.1 ± 0.5 | ~57% | 0.8 ± 0.1 | 3% |
4. Signaling Pathway Impact Analysis Mutation of the P-loop disrupts the initial step of nucleotide-dependent activation, propagating a knockout effect through the entire signaling cascade.
5. The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for P-loop SDM & Functional Studies
| Reagent/Material | Supplier Examples | Function/Application |
|---|---|---|
| High-Fidelity DNA Polymerase (Q5/Phusion) | NEB, Thermo Fisher | Error-free amplification for SDM PCR. |
| DpnI Restriction Enzyme | NEB, Thermo Fisher | Selective digestion of methylated parental plasmid template post-PCR. |
| Competent E. coli (High-Efficiency) | NEB, Agilent | Transformation of mutagenesis reaction product. |
| Site-Directed Mutagenesis Kit (e.g., QuikChange) | Agilent | Streamlined, kit-based protocol alternative. |
| [γ-³²P]ATP or [α-³²P]GTP | PerkinElmer, Hartmann Analytic | Radiolabeled nucleotides for direct binding assays. |
| Nitrocellulose Filter Membranes | MilliporeSigma, Cytiva | Capture protein-nucleotide complexes in filter-binding assays. |
| Coupled Enzyme ATPase/GTPase Assay Kit | Cytoskeleton, Sigma-Aldrich | Convenient, spectrophotometric measurement of hydrolysis kinetics. |
| Ni-NTA Agarose Resin | Qiagen, Cytiva | Immobilized metal affinity chromatography for His-tagged protein purification. |
| Fast Protein Liquid Chromatography (FPLC) System | Cytiva, Bio-Rad | High-resolution purification of proteins for structural/functional studies. |
6. Conclusion Systematic site-directed mutagenesis of P-loop residues, followed by rigorous quantitative binding and enzymatic assays, provides an unambiguous functional knockout strategy. This approach is indispensable for validating the mechanistic hypotheses concerning NBS domain function and for identifying critical residues that may serve as targets for allosteric inhibition in drug discovery programs. The data generated feeds directly into structural models and informs the understanding of disease-associated mutations in this fundamental nucleotide-handling motif.
Nucleotide-Binding Site (NBS) domains, characterized by conserved motifs such as the phosphate-binding loop (P-loop), are critical for ATP/GTP hydrolysis in proteins like kinases, GTPases, and NBS-Leucine-Rich Repeat (NLR) immune receptors. Understanding the precise molecular interactions governing nucleotide binding and hydrolysis is a central objective in structural biology and drug discovery. Computational modeling, specifically molecular docking and Molecular Dynamics (MD) simulations, provides an indispensable toolkit for probing these interactions at atomic resolution, complementing experimental techniques like X-ray crystallography and Cryo-EM. This guide details the protocols and applications of these computational methods within the broader thesis of elucidating P-loop function, dynamics, and allosteric regulation.
Core Methodologies and Experimental Protocols
Molecular Docking for Initial Pose Prediction
Molecular docking predicts the preferred orientation of a ligand (e.g., ATP, GTP, or an inhibitor) within a protein's binding pocket.
Detailed Protocol:
- System Preparation:
- Protein: Obtain the 3D structure of the NBS domain from the PDB (e.g., 3KWF for a human NLR protein). Remove water molecules and heteroatoms. Add missing hydrogen atoms and assign protonation states (e.g., using
pdb4amberorPDB2PQR). For the P-loop, ensure key residues (typically G-X-X-X-X-G-K-T/S) are in the correct state. - Ligand: Obtain the 3D structure of the nucleotide or analog (e.g., ATP-γ-S, ADP) from PubChem. Prepare using tools like
Open Babelto assign Gasteiger charges and minimize energy with MMFF94.
- Protein: Obtain the 3D structure of the NBS domain from the PDB (e.g., 3KWF for a human NLR protein). Remove water molecules and heteroatoms. Add missing hydrogen atoms and assign protonation states (e.g., using
- Define the Binding Site:
- The search space is centered on the P-loop motif. Coordinates can be derived from a known co-crystal structure or predicted from sequence alignment with a known structure.
- Perform Docking:
- Use software like AutoDock Vina or AutoDock-GPU. A sample Vina command:
- Post-docking Analysis:
- Cluster poses by RMSD. Select the top-scoring pose(s) for further analysis. Key interactions to validate include hydrogen bonds between the nucleotide phosphate(s) and the P-loop's conserved lysine and serine/threonine, and stacking interactions with aromatic residues.
Molecular Dynamics Simulations for Dynamics & Energetics
MD simulations model the time-dependent behavior of the protein-ligand complex, capturing conformational changes, binding stability, and free energy landscapes.
Detailed Protocol:
- System Building:
- Use the top docking pose. Place the complex in a TIP3P water box (e.g., with a 10 Å buffer). Add ions (e.g., Na⁺, Cl⁻) to neutralize the system charge and achieve a physiological salt concentration (e.g., 150 mM NaCl).
- Energy Minimization and Equilibration:
- Minimization: Perform 5,000-10,000 steps of steepest descent/conjugate gradient to remove steric clashes.
- Equilibration NVT: Heat the system from 0 K to 300 K over 100 ps using a Langevin thermostat, restraining protein heavy atoms.
- Equilibration NPT: Further equilibrate for 100-200 ps at 1 atm pressure (Berendsen/Parinello-Rahman barostat) with restrained protein backbone.
- Production Run:
- Run an unrestrained simulation for a timescale relevant to the biological process (typically 100 ns to 1 µs for nucleotide binding studies). Use a 2-fs integration time step. Employ particle mesh Ewald (PME) for long-range electrostatics.
- Trajectory Analysis:
- Root Mean Square Deviation (RMSD): Assess overall system stability.
- Root Mean Square Fluctuation (RMSF): Identify flexible regions (e.g., P-loop dynamics).
- Interaction Analysis: Calculate hydrogen bond occupancy, contact maps, and distance plots for key interactions (e.g., between Mg²⁺ ion, phosphates, and P-loop residues).
- Binding Free Energy: Compute using methods like MM/PBSA or MM/GBSA on trajectory frames.
Table 1: Typical MD Simulation Parameters for NBS-Nucleotide Systems
| Parameter | Typical Setting | Rationale |
|---|---|---|
| Force Field | Amber ff19SB (Protein), GAFF2 (Ligand) | Accuracy for proteins and small molecules. |
| Water Model | TIP3P | Standard, computationally efficient. |
| Box Type | Orthorhombic | Simple geometry. |
| Ion Concentration | 150 mM NaCl | Physiological relevance. |
| Temperature | 300 K | Physiological temperature. |
| Pressure | 1 atm | Isotropic-isobaric (NPT) ensemble. |
| Time Step | 2 fs | With SHAKE on bonds involving H. |
| Non-bonded Cutoff | 10-12 Å | For direct space electrostatics & VdW. |
| Long-range Electrostatics | Particle Mesh Ewald (PME) | Accurate treatment of electrostatics. |
| Production Run Length | 100 ns - 1 µs | Balance of sampling and computational cost. |
Key Research Reagent Solutions & Essential Materials
Table 2: Essential Computational Toolkit for NBS-Nucleotide Modeling
| Item | Function & Description |
|---|---|
| Protein Data Bank (PDB) | Repository for 3D structural data of proteins and nucleic acids. Source for initial NBS domain coordinates. |
| PubChem | Database of chemical molecules and their activities. Source for nucleotide and inhibitor 3D structures. |
| CHARMM/Amber Force Fields | Parameter sets defining potential energy functions for atoms in the simulation (bonds, angles, dihedrals, electrostatics, VdW). |
| AutoDock Vina/GOLD | Widely used molecular docking programs for predicting ligand-receptor binding modes and affinities. |
| GROMACS/AMBER/NAMD | High-performance MD simulation software packages for running energy minimization, equilibration, and production MD. |
| Visual Molecular Dynamics (VMD) | Molecular visualization and analysis program for displaying trajectories, rendering images, and basic analysis. |
| PyMOL | Molecular graphics system for high-quality rendering and presentation of protein-ligand structures. |
| Python (MDanalysis, MDAnalysis) | Programming language with specialized libraries for scripting, analyzing, and visualizing MD trajectory data. |
| High-Performance Computing (HPC) Cluster | Essential computational resource (CPUs/GPUs) for running MD simulations, which are highly computationally intensive. |
Workflow and Analysis Visualization
Title: Computational Modeling Workflow for Nucleotide Interactions
Title: Key Steps in P-loop Mediated ATP Hydrolysis
Quantitative Data and Analysis
Table 3: Example MM/GBSA Binding Free Energy Results for ATP Analogs to an NBS Domain
| Ligand | ΔE_vdW (kcal/mol) | ΔE_elec (kcal/mol) | ΔG_GB (kcal/mol) | ΔG_SA (kcal/mol) | ΔG_Total (kcal/mol) | Key Interacting P-loop Residue |
|---|---|---|---|---|---|---|
| ATP | -45.2 ± 3.1 | -225.5 ± 15.2 | 240.1 ± 14.8 | -6.2 ± 0.4 | -36.8 ± 2.5 | Lys189, Ser190 |
| ADP | -40.1 ± 2.8 | -180.3 ± 12.4 | 195.5 ± 13.1 | -5.8 ± 0.4 | -30.7 ± 2.1 | Lys189 |
| ATP-γ-S | -46.5 ± 3.3 | -218.9 ± 14.7 | 235.8 ± 15.0 | -6.5 ± 0.5 | -36.1 ± 2.6 | Lys189, Ser190 |
Table 4: Hydrogen Bond Occupancy (%) from a 200 ns MD Simulation (ATP-bound NBS)
| Donor | Acceptor | Occupancy (%) | Role in Catalysis |
|---|---|---|---|
| P-loop Lys (NZ) | ATP α-Phosphate (O) | 98.5 | Critical for phosphate positioning |
| P-loop Ser (OG) | ATP γ-Phosphate (O) | 85.2 | Stabilizes transferring phosphate |
| Walker B Asp (OD1) | Mg²⁺ ion | 99.9 | Mg²⁺ coordination |
| Mg²⁺ (ion) | ATP β,γ-Phosphates (O) | 99.5 | Substrate activation |
Computational modeling via docking and MD simulations is a powerful paradigm for dissecting the mechanistic details of nucleotide interaction with NBS domain P-loops. By providing dynamic and energetic insights complementary to static structures, these methods can identify key conformational states, allosteric networks, and the impact of disease-associated mutations. Integration with enhanced sampling techniques (e.g., metadynamics) and machine learning will further accelerate the discovery of selective inhibitors targeting NBS domains in kinases, GTPases, and NLR proteins for therapeutic intervention.
This whitepaper is framed within a broader thesis investigating the function of conserved motifs, specifically the Phosphate-binding loop (P-loop), within the Nucleotide-Binding Site (NBS) domains of proteins. The P-loop, with a canonical sequence of G-X-X-X-X-G-K-T/S, is a critical structural element found in numerous enzyme families, including kinases, GTPases, and ATP-binding cassette (ABC) transporters. Its primary function is to coordinate the phosphate groups of nucleotides (e.g., ATP, GTP), facilitating essential biological processes such as signal transduction, cytoskeletal regulation, and active transport.
Dysregulation of P-loop-containing proteins is implicated in a wide array of diseases, including cancer, autoimmune disorders, and neurodegenerative conditions. Mutations within the P-loop can alter nucleotide binding affinity or hydrolysis, leading to constitutive activation or inactivation of the protein. Consequently, the P-loop presents a compelling, structurally conserved target for rational drug design. This guide provides an in-depth technical analysis of strategies to target this motif, supported by current experimental data and protocols.
Quantitative Data on Disease-Associated P-loop Proteins
The following tables summarize key quantitative data on prominent disease-associated proteins featuring a critical P-loop.
Table 1: Oncogenic Kinases with Targetable P-loops
| Protein Name (Gene) | Disease Association | Canonical P-loop Sequence | Reported Kd for ATP (μM) | Common P-loop Mutations & Effect |
|---|---|---|---|---|
| B-Raf (BRAF) | Melanoma, CRC | G-S-S-G-E-G-K-T | ~15 | V600E: Increases kinase activity >500-fold. |
| c-Kit (KIT) | GIST, AML | G-E-G-G-G-K-T | ~10 | D816V: Constitutive activation, resistance. |
| EGFR (EGFR) | NSCLC, GBM | G-S-F-G-E-V-K-T | ~5 | L858R: Increases ATP affinity 10-20x. |
| ABL1 (ABL1) | CML, ALL | G-E-G-G-W-G-K-T | ~0.6 | T315I (gatekeeper): Blocks inhibitor binding. |
Table 2: GTPases and Other P-loop Proteins in Disease
| Protein Class/Name | Disease Association | Nucleotide | Key P-loop Interaction | Inhibitor Strategy Example |
|---|---|---|---|---|
| K-Ras (KRAS) | Pancreatic, CRC, NSCLC | GTP/GDP | G-A-G-G-V-G-K-S | Direct covalent inhibitors (e.g., sotorasib). |
| RhoA (RHOA) | Cancer metastasis | GTP/GDP | G-D-P-G-G-K-T | Targeting downstream effectors. |
| Nucleotide-Binding Domain (NBD) of CFTR | Cystic Fibrosis | ATP | L-S-G-G-Q-Q-K-T | Potentiators (e.g., ivacaftor) stabilize open state. |
| NLRP3 (NACHT domain) | Inflammasome disorders | ATP/dATP | G-Y-P-G-T-G-K-T | MCC950 binds to Walker B motif, blocks ATP hydrolysis. |
Experimental Protocols for P-loop Targeting
Protocol: In Silico Screening for P-loop Binders
Objective: Identify small molecules that preferentially bind to the P-loop conformation of a target protein.
- Protein Preparation: Retrieve target structure (e.g., PDB ID: 1MV5 for B-Raf) or generate a homology model. Add missing hydrogen atoms, assign protonation states (pH 7.4), and optimize side-chain conformations using software like Schrödinger's Protein Preparation Wizard or UCSF Chimera.
- P-loop Site Definition: Define the binding pocket as residues within 8-10 Å of the phosphate groups of the co-crystallized ATP/ANP or the Mg2+ ions.
- Ligand Library Preparation: Prepare a library of 50,000-500,000 drug-like molecules (e.g., ZINC15, Enamine). Generate 3D conformers and minimize energies using force fields like MMFF94s.
- Virtual Screening: Perform high-throughput docking (HTD) using GLIDE SP mode or AutoDock Vina. Prioritize compounds based on docking score, favorable interactions with P-loop backbone amides (Gly, Lys), and phosphate-mimicking groups (e.g., sulfonamides, carboxylates).
- Post-Docking Analysis: Cluster top 1000 hits. Visually inspect for key hydrogen bonds with the invariant Lys and Ser/Thr residues. Perform MM-GBSA calculations on top 100 hits for binding free energy estimation.
Protocol: Surface Plasmon Resonance (SPR) for Binding Kinetics
Objective: Determine the real-time binding kinetics (KD, kon, koff) of a lead compound to the purified P-loop protein.
- Immobilization: Dilute recombinant, purified target protein (≥95% purity) in 10 mM sodium acetate buffer (pH 5.0). Using a Biacore T200 system, immobilize the protein on a CM5 sensor chip via amine coupling to achieve a response unit (RU) increase of 8,000-12,000.
- Ligand Preparation: Serially dilute the lead compound in running buffer (e.g., HBS-EP+: 10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.05% P20 surfactant, pH 7.4). Include a DMSO control (match final DMSO ≤1%).
- Binding Assay: Use a multi-cycle kinetics method. Inject compound dilutions (0.1 nM - 10 μM) over the protein and reference surfaces at a flow rate of 30 μL/min for 120s association time, followed by 300s dissociation time.
- Data Analysis: Subtract reference and buffer injection signals. Fit the resulting sensorgrams to a 1:1 binding model using the Biacore Evaluation Software to calculate association (kon) and dissociation (koff) rate constants. Calculate equilibrium dissociation constant KD = koff/kon.
Protocol: Cellular Thermal Shift Assay (CETSA)
Objective: Validate target engagement of a P-loop inhibitor in a live-cell context.
- Cell Treatment: Culture relevant cell lines (e.g., A375 for BRAF V600E). Treat with compound or DMSO vehicle for 2 hours at desired concentrations.
- Heat Challenge: Harvest cells, wash with PBS, and resuspend in PBS with protease inhibitors. Aliquot equal volumes (~50 μL) into PCR tubes. Heat each aliquot at a range of temperatures (e.g., 37°C to 65°C, 3°C increments) for 3 minutes in a thermal cycler.
- Lysate Preparation: Freeze-heat-thaw cycles: Freeze samples in liquid nitrogen for 2 minutes, then thaw at room temperature. Repeat twice. Centrifuge at 20,000 x g for 20 minutes at 4°C to pellet aggregated protein.
- Western Blot Analysis: Transfer supernatants to new tubes. Perform SDS-PAGE and Western blotting for the target protein (e.g., BRAF) and a loading control (e.g., GAPDH). Quantify band intensity.
- Data Interpretation: Calculate the percentage of soluble protein remaining at each temperature. A leftward shift in the melting curve (Tm) for the compound-treated sample indicates thermal stabilization and direct target engagement.
Visualizations
Diagram 1: P-loop in NBS Domain Signaling Context
P-loop in Nucleotide Binding and Activation
Diagram 2: Workflow for P-loop Drug Discovery
P-loop Inhibitor Discovery Pipeline
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for P-loop Research
| Reagent/Material | Function in P-loop Research | Example Product/Source |
|---|---|---|
| Recombinant P-loop Protein (Wild-type & Mutant) | Essential for structural studies, biochemical assays (SPR, enzymatic), and crystallography for drug screening. | Purified human KRAS G12C (Cytoskeleton, Inc.); Recombinant NLRP3 NACHT domain (Novus Biologicals). |
| ATPase/GTPase Activity Assay Kit | Measures the functional output of P-loop nucleotide hydrolysis; critical for inhibitor validation. | Colorimetric ATPase Assay Kit (Innova Biosciences); GTPase-Glo Assay (Promega). |
| Cellular Thermal Shift Assay (CETSA) Kit | Validates target engagement of inhibitors in a physiologically relevant cellular environment. | CETSA Cellular Thermal Shift Assay Kit (Thermo Fisher Scientific). |
| Anti-Phospho-Substrate Antibodies | Detects downstream pathway activation/inhibition (e.g., p-ERK for RAF inhibition). | Phospho-p44/42 MAPK (Erk1/2) (Thr202/Tyr204) Antibody (Cell Signaling Technology). |
| P-loop Directed Covalent Probe | Chemical probes for occupancy studies and target validation via chemoproteomics. | Sotorasib (AMG 510) derivative probe for KRAS G12C. |
| Surface Plasmon Resonance (SPR) Chip | For label-free kinetic analysis of small molecule binding to immobilized P-loop proteins. | Series S Sensor Chip CM5 (Cytiva). |
| Nucleotide Analogs (ATP-γ-S, GTP-γ-S) | Hydrolysis-resistant analogs for structural studies to trap P-loop conformation. | Adenosine 5'-[γ-thio]triphosphate (ATPγS) (Jena Bioscience). |
| Molecular Modeling/Docking Software Suite | For in silico screening and analysis of P-loop inhibitor interactions. | Schrödinger Suite; AutoDock Vina. |
Challenges in P-loop Analysis: Overcoming Experimental Pitfalls and Artifacts
This technical guide addresses critical experimental challenges within a broader thesis investigating the function of conserved motifs, specifically the Phosphate-binding loop (P-loop), in Nucleotide-Binding Site (NBS) domains. The P-loop (GxxxxGK[T/S]) is a quintessential Walker A motif essential for coordinating phosphate moieties of ATP or GTP. Research into its mechanistic role in nucleotide hydrolysis, conformational switching, and signal transduction in kinases, GTPases, and NLR immune receptors is often hindered by the inherent instability of purified NBS domain proteins and the complexities of accurately measuring their nucleotide affinity and kinetics. This whitepaper provides an in-depth analysis of these common issues and offers optimized, actionable protocols for robust experimentation.
Part 1: Protein Stability Challenges and Solutions
Core Stability Issues in NBS Domain Proteins
NBS domain proteins, especially when expressed as isolated domains, frequently exhibit poor solubility, aggregation, and loss of cofactor-dependent structural integrity.
Table 1: Common Protein Stability Issues and Their Impact on P-loop Studies
| Issue | Primary Cause | Impact on P-loop Function Assay |
|---|---|---|
| Low Expression Yield | Poor solubility, codon bias, toxicity. | Insufficient material for titration experiments. |
| Aggregation During Purification | Exposed hydrophobic surfaces, lack of ligand. | P-loop conformation obscured; non-specific binding. |
| Rapid Activity Loss | Oxidation of cysteines, metal cofactor dissociation. | Inaccurate measurement of ATPase kinetics. |
| Conformational Heterogeneity | Flexible switch regions (Walker B, Sensor-1) without nucleotide. | High background in binding assays; poor crystallography. |
Optimized Protocol for Stabilizing NBS Domain Proteins
Methodology: Ligand-Stabilized Expression and Purification
- Construct Design: Include a solubility tag (e.g., MBP, GST). Clone the NBS domain with defined boundaries from bioinformatics analysis (often ~150-300 residues around the P-loop).
- Expression: Use E. coli BL21(DE3) pLysS or insect cell systems for mammalian NBS proteins. Induce at low temperature (18-22°C for E. coli, 27°C for insect cells) with 0.1-0.5 mM IPTG or at OD600 ~0.6-0.8.
- Critical Stabilization Step: Supplement growth media and all purification buffers with 10 mM MgCl₂ and 1-5 mM non-hydrolyzable nucleotide analog (e.g., AMP-PNP, ADP, GDP). This maintains the P-loop in a ligand-bound, folded state.
- Purification: Perform IMAC (if His-tagged) in Buffer A: 50 mM HEPES pH 7.5, 300 mM NaCl, 10 mM MgCl₂, 5% glycerol, 1 mM TCEP, 1 mM AMP-PNP. Use a gradient elution with imidazole (20-500 mM).
- Size-Exclusion Chromatography (SEC): Use a HiLoad 16/600 Superdex 200 pg column equilibrated in Buffer A without imidazole but with 0.5-1 mM nucleotide analog. Collect the monodisperse peak.
- Storage: Flash-freeze in small aliquots in liquid nitrogen with 10% glycerol. Avoid repeated freeze-thaw cycles.
Part 2: Nucleotide Binding Assay Optimization
Challenges in Quantifying P-loop Ligand Interactions
Traditional methods like filter binding or radioactive assays pose safety and waste issues. Fluorescence-based methods can be affected by protein instability or non-specific quenching.
Table 2: Comparison of Nucleotide Binding Assay Techniques
| Assay Type | Principle | Key Advantage | Key Limitation for NBS Domains | Typical Kd Range |
|---|---|---|---|---|
| Isothermal Titration Calorimetry (ITC) | Measures heat of binding. | Yields Kd, ΔH, ΔS, stoichiometry (n) in solution. | Requires high protein concentration and stability. | μM to nM |
| MicroScale Thermophoresis (MST) | Tracks fluorescence changes due to temperature-induced motion. | Low sample consumption; works in complex buffers. | Sensitive to fluorescence interference. | nM to mM |
| Fluorescence Polarization/Anisotropy (FP/FA) | Measures change in rotation speed of a fluorescent ligand. | Homogeneous, real-time, high-throughput. | Requires fluorescent nucleotide analogs which may alter affinity. | nM to μM |
| Surface Plasmon Resonance (SPR) | Measures mass change on a sensor chip surface. | Provides real-time kinetics (ka, kd). | Risk of measuring avidity if protein is multimeric; requires immobilization. | nM to μM |
Optimized Protocol: ITC for NBS Domain Nucleotide Binding
ITC remains the gold standard for label-free, in-solution binding characterization.
Detailed ITC Methodology:
Sample Preparation:
- Protein: Dialyze the purified, stabilized NBS domain extensively against ITC Buffer: 25 mM HEPES pH 7.5, 150 mM NaCl, 10 mM MgCl₂, 1 mM TCEP. Degas for 10 minutes prior to loading.
- Ligand: Dissolve ATP, ADP, or analog (e.g., ATPγS) directly in the final dialysis buffer from the protein sample. Match pH precisely.
Instrument Setup (e.g., Malvern MicroCal PEAQ-ITC):
- Cell: Fill with NBS protein at 10-50 μM concentration (monomer).
- Syringe: Fill with nucleotide at 10-20x the cell concentration (e.g., 200-500 μM).
- Temperature: 25°C.
- Reference Power: 5-10 μCal/sec.
- Stirring Speed: 750 rpm.
Titration Program:
- Initial delay: 60 sec.
- Number of injections: 19-25.
- Injection volume: 1.5-2.0 μL (first injection can be 0.5 μL discarded).
- Duration per injection: 3-4 sec.
- Spacing between injections: 150-180 sec.
Data Analysis:
- Integrate raw heat peaks.
- Subtract control titration (nucleotide into buffer).
- Fit integrated data to a "One Set of Sites" binding model using the instrument software.
- Critical Parameters: Ensure stoichiometry (N) is close to 1.0 for a monomeric binding event. A poor fit may indicate protein instability or aggregation during the run.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for NBS Domain P-loop Studies
| Reagent | Function in Experiment | Key Consideration |
|---|---|---|
| Non-hydrolyzable Nucleotides (AMP-PNP, ATPγS, GMP-PNP) | Stabilizes P-loop conformation during purification; used as ligands in binding assays. | ATPγS is a closer mimic to ATP than AMP-PNP but can be slowly hydrolyzed by some enzymes. |
| TCEP (Tris(2-carboxyethyl)phosphine) | Reducing agent to prevent disulfide formation in cysteine-rich NBS domains. | More stable than DTT in buffer; use at 0.5-2 mM. |
| HEPES Buffer (pKa 7.5) | Maintains physiological pH with minimal metal chelation. | Preferred over Tris for assays involving Mg²⁺ ions. |
| MgCl₂ | Essential divalent cation for P-loop coordination of nucleotide phosphates. | Concentration (5-20 mM) is critical; excess can promote non-specific binding. |
| Size-Exclusion Chromatography Standards | To determine oligomeric state and monodispersity of purified protein. | Run with and without nucleotide to assess ligand-induced oligomerization. |
| Fluorescent Nucleotide Analogs (e.g., Mant-ATP, TNP-ATP) | For FP/FA, MST, or stopped-flow kinetics assays. | The fluorophore can significantly alter binding affinity; always validate with a label-free method. |
Visualizing the Workflow and Signaling Context
Diagram Title: NBS Domain Protein Study Workflow from Gene to Thesis
Diagram Title: NLR Immune Receptor Activation via P-loop Nucleotide Exchange
Successful research into the conserved function of the NBS domain P-loop motif is predicated on overcoming dual hurdles of protein stability and assay fidelity. By implementing ligand-stabilized purification protocols and selecting the appropriate binding assay with careful optimization, researchers can generate robust, quantitative data. This data, revealing precise thermodynamic and kinetic parameters of nucleotide interaction, forms the experimental foundation for advancing the mechanistic thesis on how P-loop conformational dynamics govern signaling across diverse protein families.
The P-loop (Phosphate-binding loop), a canonical Walker A motif (GxxxxGK[S/T]), is a universal nucleotide-binding module critical for function in nucleotide-binding site (NBS) domain proteins, including kinases, GTPases, and AAA+ ATPases. A core challenge in mechanistic studies of these proteins arises when a point mutation within this motif results in a loss-of-function phenotype. The pivotal question for interpreting such a mutant is: does the defect originate from an inability to bind the nucleotide (ATP/GTP) or an inability to hydrolyze it post-binding? Distinguishing between these two classes of defects is fundamental to understanding molecular mechanism, informing disease pathogenesis (e.g., mutations in kinases or RAS family proteins), and guiding rational drug design. This guide details the experimental framework for making this critical distinction.
Table 1: Key Assays for Distinguishing Binding vs. Hydrolysis Defects
| Assay Category | Specific Assay | Measures | Interpretation for P-loop Mutant |
|---|---|---|---|
| Direct Binding | Isothermal Titration Calorimetry (ITC) | Binding affinity (KD), enthalpy (ΔH) | ↑ KD (weaker binding) = Binding defect. Normal KD suggests defect elsewhere. |
| Surface Plasmon Resonance (SPR) | Association/dissociation rates (kon, koff), KD | ↑ koff (faster dissociation) = Binding defect. | |
| Fluorescence Polarization/Anisotropy | KD using labelled nucleotide | ↑ KD = Binding defect. | |
| Hydrolysis-Coupled | Malachite Green Phosphate Assay | Inorganic phosphate (Pi) release over time | Low Pi with normal binding (from Table 2) = Hydrolysis defect. |
| NADH-Coupled Enzymatic Assay | ATP consumption via absorbance at 340 nm | Low ATPase rate with normal binding = Hydrolysis defect. | |
| Structural & Stabilization | Thermal Shift Assay (TSA) | Protein melting temperature (Tm) ± nucleotide | Nucleotide-induced ΔTm lost = Binding defect. Persistent ΔTm = binding intact. |
| Tryptophan Fluorescence Quenching | Conformational change upon nucleotide addition | Loss of quenching signal = Binding or allosteric defect. |
Table 2: Hypothetical Data for a P-loop Lysine Mutant (K→A)
| Protein | KD (ATP) (ITC) | kcat (Hydrolysis) | Tm (°C) (Apo) | Tm (°C) (+ATP) | ΔTm | Interpretation |
|---|---|---|---|---|---|---|
| Wild-Type | 10 µM | 100 min-1 | 45.0 | 52.5 | +7.5 | Baseline |
| Mutant K→A | >500 µM | Not detectable | 44.5 | 44.8 | +0.3 | Severe binding defect |
| Mutant S→A | 12 µM | 1.5 min-1 | 45.2 | 52.0 | +6.8 | Catalytic hydrolysis defect |
Detailed Experimental Protocols
Protocol: Isothermal Titration Calorimetry (ITC) for Nucleotide Binding
Objective: Determine the thermodynamic parameters of nucleotide binding to the wild-type versus P-loop mutant protein.
- Sample Preparation: Dialyze purified protein (in 20 mM HEPES pH 7.5, 150 mM NaCl, 5 mM MgCl2) against assay buffer. Dissolve ATP (or GTP) in the final dialysis buffer.
- Instrument Setup: Load the protein solution (50-100 µM) into the sample cell. Fill the syringe with nucleotide solution (10x concentrated relative to protein).
- Titration: Perform a series of injections (e.g., 19 x 2 µL) with stirring. The instrument measures heat released/absorbed after each injection.
- Data Analysis: Integrate raw heat peaks. Fit the binding isotherm to an appropriate model (e.g., one-set-of-sites) to derive KD, stoichiometry (N), enthalpy (ΔH), and entropy (ΔS).
Protocol: Malachite Green Phosphate Release Assay
Objective: Quantify the hydrolytic activity of the protein-nucleotide complex over time.
- Reaction Setup: In a 96-well plate, mix protein (wild-type or mutant, 1 µM final) in reaction buffer (e.g., 40 mM Tris-HCl pH 7.5, 80 mM NaCl, 10 mM MgCl2) with ATP (1 mM final). Incubate at desired temperature.
- Time-Course Sampling: At intervals (e.g., 0, 2, 5, 10, 20, 30 min), remove an aliquot and quench it with an equal volume of Malachite Green reagent (0.045% malachite green, 4.2% ammonium molybdate in 4N HCl, with Tween-20).
- Detection & Quantification: After 1-5 minutes at room temperature, measure absorbance at 620 nm. Compare to a standard curve of known phosphate concentrations (KH2PO4).
- Kinetics: Plot Pi released vs. time. The initial slope gives the hydrolysis rate. A mutant with wild-type binding but severely reduced rate indicates a hydrolysis defect.
Protocol: Thermal Shift Assay (Differential Scanning Fluorimetry)
Objective: Assess if nucleotide binding stabilizes the protein, indicating successful complex formation.
- Plate Setup: In a real-time PCR tube/plate, mix 10 µL of protein solution (5 µM) with 10 µL of buffer ± nucleotide (e.g., 1 mM ATP). Include a fluorescent dye (e.g., SYPRO Orange, 5X final).
- Thermal Ramp: Run a thermal ramp from 25°C to 95°C with a gradual increase (e.g., 1°C/min) while monitoring fluorescence.
- Data Analysis: Plot fluorescence vs. temperature. The inflection point (Tm) is where the protein unfolds and dye binds exposed hydrophobes. Calculate ΔTm = Tm(+nucleotide) - Tm(apo). A loss of ΔTm in a mutant suggests a binding defect.
Visualizations
Title: Decision Workflow for Interpreting P-loop Mutants
Title: Catalytic Cycle with Mutant Block Points
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for P-loop Functional Analysis
| Reagent / Material | Function in Analysis | Key Consideration |
|---|---|---|
| High-Purity Nucleotides (ATP, GTP, analogs) | Substrate for binding & hydrolysis assays. | Use ultrapure, HPLC-grade. Include Mg²⁺ chelating salts. Consider non-hydrolyzable analogs (AMP-PNP, GMP-PNP) for pure binding studies. |
| ITC / SPR Instrumentation | Gold-standard for quantitative binding kinetics & thermodynamics. | Requires significant protein (>0.5 mg) at high concentration. Buffer matching is critical to minimize heat of dilution (ITC). |
| Malachite Green Phosphate Assay Kit | Colorimetric detection of inorganic phosphate (Pi). | Sensitive to free phosphate contamination. Must run a standard curve with each experiment. |
| Thermal Shift Dye (e.g., SYPRO Orange) | Binds hydrophobic patches exposed during protein thermal denaturation. | Optimize dye:protein ratio. Dye can sometimes interfere with protein function; validate. |
| Stable Isotope-Labeled Nucleotides (γ-³²P-ATP, α-³²P-GTP) | Radiolabel for ultra-sensitive activity assays (TLC, charcoal-binding). | Enables measurement of low residual activity in severe mutants. Requires radiation safety protocols. |
| Crystallography Screens (e.g., Hampton Research) | To solve structures of mutant-nucleotide complexes. | Definitive proof of binding mode. Apo and bound structures reveal conformational consequences. |
The P-loop (phosphate-binding loop), a canonical glycine-rich motif (GXXXXGK[T/S]) within nucleotide-binding sites (NBS), is fundamental to the function of ATPases, GTPases, and kinases. Its primary role is to coordinate the phosphate moiety of nucleotides, a process critically dependent on the presence of a divalent cation, typically Mg²⁺. Structural biology techniques—X-ray crystallography, cryo-electron microscopy (cryo-EM), and NMR—are indispensable for elucidating the mechanistic details of P-loop mediated catalysis and regulation. However, the interpretation of these structures is fraught with potential artifacts, particularly concerning the identity and occupancy of the bound cation and the use of non-hydrolyzable ATP analogues. Misidentification of a bound metal ion (e.g., Na⁺ vs. Mg²⁺) or misinterpretation of an analogue's binding mode can lead to incorrect mechanistic models. This guide provides a technical framework for designing and validating structural studies of NBS domains to avoid such pitfalls, ensuring data integrity for downstream drug discovery targeting these ubiquitous motifs.
The Central Role of Mg²⁺ and Common Artifacts
Mg²⁺ is not merely a passive cofactor; it is an integral catalytic participant. It neutralizes negative charge, polarizes the γ-phosphate for nucleophilic attack, and stabilizes transition states. In structural studies, artifacts arise from several sources:
- Ion Identity Ambiguity: Mg²⁺ (ionic radius ~0.72 Å), Mn²⁺ (~0.83 Å), and even Na⁺ (~1.02 Å) can appear as similar electron density spheres at moderate resolutions (>2.0 Å). Soaking or co-crystallization with high, non-physiological concentrations of cations from crystallization buffers can lead to mis-assignment or spurious binding sites.
- Coordination Geometry Neglect: Mg²⁺ exhibits a strong preference for octahedral coordination with oxygen ligands (water or protein carboxylates). A metal site with distorted geometry or fewer than six ligands should be scrutinized.
- pH-Dependent Occupancy: Mg²⁺ binding affinity to ATP and the P-loop is pH-sensitive. Structures solved at non-physiological pH may show altered or absent metal ion occupancy.
Table 1: Common Divalent Cations in Structural Studies: Properties and Ambiguities
| Cation | Ionic Radius (Å) | Preferred Coordination Geometry | Common X-ray Anomalous Signal (Edge, keV) | Risk of Misassignment as Mg²⁺ |
|---|---|---|---|---|
| Mg²⁺ | 0.72 | Octahedral | Very weak (Na, 1.30) | Reference standard |
| Mn²⁺ | 0.83 | Octahedral | Strong (Kα, 6.54) | High (similar size/geometry) |
| Ca²⁺ | 1.00 | Variable (6-8) | Weak (Kα, 4.04) | Medium (if geometry ignored) |
| Na⁺ | 1.02 | Variable (4-6) | None | High at low resolution |
ATP Analogues: Strategic Use and Inherent Limitations
Non-hydrolyzable or ground-state ATP analogues are essential for trapping pre-hydrolysis states. Each has specific properties that can introduce structural artifacts if not properly considered.
Table 2: Common ATP Analogues and Their Structural Implications
| Analogue | Key Modification | Intended Trapped State | Potential Artifacts & Considerations |
|---|---|---|---|
| AMP-PNP | NH replaces O between β- and γ-P | Pre-hydrolysis (ground state) | Altered metal coordination (Mg²⁺ binds weakly to NH), can adopt non-productive conformations. |
| ATPγS | O replaced by S at γ-P | Pre-hydrolysis / Transition state | Altered metal affinity (thiophosphate is a softer ligand), may resist phosphorylation but can sometimes be slowly hydrolyzed. |
| ADP·BeF₃⁻ | BeF₃⁻ mimics γ-PO₄⁻ | Post-hydrolysis transition state mimic | Highly accurate. BeF₃⁻ geometry perfectly mimics PO₄. Toxic. Requires careful handling. |
| ADP·AlF₄⁻ / ADP·AlF₃ | AlF₄⁻ mimics PO₄⁻ in pentavalent transition state; AlF₃ mimics γ-PO₄⁻ | Pentavalent transition state (AlF₄⁻) or ground state (AlF₃) | Powerful mimics but AlFx complexes can adopt multiple coordination modes. |
| ADP·VO₄ | VO₄ mimics PO₄⁻ in pentavalent transition state | Transition state mimic | Vanadate can form dimers or adopt distorted geometries. |
Experimental Protocols for Artifact Avoidance
Protocol: Validating Metal Ion Identity in Crystallography
- Data Collection: Collect a high-resolution native dataset (≤1.8 Å ideal) to visualize coordination geometry.
- Anomalous Scattering: Collect multi-wavelength anomalous diffraction (MAD) data at the Mn/K-edge (if Mn²⁺ is suspected) or at a long wavelength (~1.5-2.0 Å) to enhance the weak anomalous signal from Mg²⁺ (Na edge) or P/S in nucleotides.
- Refinement: Refine the metal ion with partial occupancy. Restrain its coordination geometry (bond lengths, angles) to expected values for Mg²⁺ octahedra. Use B-factor (atomic displacement parameter) analysis: a B-factor significantly different from surrounding ligands suggests misassignment.
- Validation: Check the site using the CheckMyMetal (CMM) server or equivalent tools, which compare geometry against known metalloprotein structures.
Protocol: Cryo-EM Sample Preparation for Mg²⁺-Dependent Complexes
- Buffer Optimization: Use a physiologically relevant buffer (e.g., 20 mM HEPES pH 7.5, 150 mM KCl, 1-5 mM MgCl₂). Avoid high concentrations of chelators (EDTA, citrate) or competing ions (e.g., >200 mM NaCl).
- Grid Preparation: Apply sample to grids pre-treated with a thin continuous carbon film or ultra-foil. Use a blot force and time that prevents excessive buffer dilution or crystallization of salts at the grid interface.
- Blotting/Washing: For glycerol or detergent-containing samples, a brief (5-10 sec) wash with a metal-free but otherwise identical buffer immediately before blotting can reduce background, but may risk stripping the metal cofactor. This step requires empirical optimization.
Protocol: Trapping a Pre-Hydrolysis State with AMP-PNP
- Protein Preparation: Purify the target NBS protein in a nucleotide-free state using size-exclusion chromatography in metal-free buffer + 1 mM EDTA, followed by buffer exchange into Mg²⁺-containing assay buffer.
- Complex Formation: Incubate protein with a 1.2-1.5 molar excess of AMP-PNP and a stoichiometric (1:1 with nucleotide) or slight excess (e.g., 2 mM) of MgCl₂ for 30 minutes on ice.
- Stabilization: Add a stabilizing agent if needed (e.g., 5% glycerol, 0.5 mM TCEP) before proceeding to crystallization trials or grid freezing.
- Control: Always prepare a parallel sample with authentic ATP under identical conditions to assess activity and validate that the analogue is indeed blocking hydrolysis.
Visualization: Experimental Workflow & P-loop Coordination
Diagram 1: Workflow for Trapping NBS States with Analogues
Diagram 2: Octahedral Mg²⁺ Coordination in the P-loop Active Site
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Robust NBS Structural Studies
| Reagent / Material | Function & Rationale | Key Considerations |
|---|---|---|
| Ultrapure MgCl₂ (≥99.99%) | Provides the essential cofactor without contaminating transition metals (Mn²⁺, Ni²⁺) that could mis-assign density. | Use stock solutions in Chelex-treated water. Avoid MgSO₄ if sulfate interferes with crystallization. |
| Non-hydrolyzable ATP Analogues (AMP-PNP, ATPγS) | Trap nucleotide-bound states for structure determination. | AMP-PNP: Check solubility at neutral pH. ATPγS: Source as stable lithium or sodium salts. |
| Transition State Mimics (BeF₃⁻, AlF₄⁻) | Form stable complexes with ADP to mimic the phosphoryl transfer transition state. | Highly toxic (especially Be²⁺). Handle in a fume hood with PPE. Prepare fresh from NaF + BeCl₂ or AlCl₃. |
| Chelating Resins (Chelex-100) | Remove trace divalent cations from buffers to prepare truly metal-free protein for controlled reconstitution experiments. | Column must be pre-equilibrated to correct pH. Can also bind protein; use for buffer treatment only. |
| Crystallography Grade PEGs & Salts | Precipitants for growing ordered crystals of NBS domains. | May contain trace metals. Consider passing stock solutions through Chelex columns for sensitive studies. |
| Holey Carbon Gold Grids (R1.2/1.3, 300 mesh) | Preferred support film for high-resolution cryo-EM of soluble NBS proteins. | Grid batch variability is high. Test multiple batches for optimal ice thickness and particle distribution. |
| Lathanide Salts (e.g., Yb³⁺, Lu³⁺) | Provide strong anomalous scattering signals for definitively identifying and phasing metal sites in crystallography. | Can displace native Mg²⁺. Use at low concentrations (0.1-1 mM) as a spectroscopic probe, not a substitute. |
Nucleoside-binding site (NBS) domains, characterized by the conserved phosphate-binding loop (P-loop or Walker A motif), are central to nucleotide hydrolysis in ATPases and GTPases. This guide addresses suboptimal enzymatic hydrolysis rates, a critical issue in functional studies of these motifs. Within the broader thesis on P-loop evolution and mechanism, understanding cofactor dependency and allosteric networks is paramount for interpreting in vitro kinetics and guiding drug discovery targeting nucleotide-binding proteins.
Core Regulatory Mechanisms Impacting Hydrolysis
Hydrolysis rates in P-loop NTPases are seldom intrinsic. They are finely tuned by:
- Cofactors: Essential ions (e.g., Mg²⁺, Mn²⁺) that stabilize transition states and orient reactants.
- Allosteric Regulators: Remote binding events that induce conformational shifts in the catalytic core, altering substrate affinity or catalytic efficiency.
- Interactive Networks: Communication between the P-loop, Walker B motif, and sensor motifs, often modulated by regulators.
Diagram Title: Cofactor & Allosteric Regulation of P-loop Hydrolysis
Systematic Troubleshooting Framework
Table 1: Common Cofactors and Their Typical Effects on P-loop NTPase Hydrolysis
| Cofactor | Typical Concentration Range | Primary Role | Impact on kcat (vs. no cofactor) | Notes |
|---|---|---|---|---|
| Mg²⁺ | 0.5-10 mM | Catalytic metal; stabilizes γ-phosphate & attacking nucleophile. | 10² - 10⁵ fold increase | Physiological ion; concentration is often matched to [NTP]. |
| Mn²⁺ | 0.1-2 mM | Alternative catalytic metal; often enhances kcat but reduces KM. | Variable, can exceed Mg²⁺ | Useful for mechanistic studies; may promote promiscuity. |
| Ca²⁺ | 0.1-5 mM | Inhibitory or weakly active vs. Mg²⁺; may act as a regulatory signal. | 0.01 - 0.5 fold (of Mg²⁺ rate) | Binds with higher affinity, can block Mg²⁺ site. |
| K⁺ / NH₄⁺ | 50-200 mM | Monovalent cation; stabilizes active conformation (e.g., in G proteins). | 2 - 100 fold increase | Allosteric rather than catalytic; optimizes switch I/II orientation. |
Table 2: Diagnostic Experiments for Low Hydrolysis Rates
| Suspected Issue | Experimental Test | Expected Outcome if Issue is Present | Key Controls |
|---|---|---|---|
| Insufficient/Incorrect Cofactor | Titrate Mg²⁺ (0-20 mM) with constant [ATP]. | Rate peaks then declines; optimum often at ~1:1 Mg:ATP. | Include EDTA-treated enzyme; test Mn²⁺. |
| Missing Allosteric Activator | Add known protein partners, regulatory nucleotides (e.g., GAPs for GTPases). | Rate increases sharply upon addition. | Use catalytically dead mutant partner. |
| Inhibitory Divalent Cations | Replace Mg²⁺ with Ca²⁺ or include Zn²⁺ (0.1 mM). | Rate is severely diminished. | Measure with pure Mg²⁺ system. |
| Non-optimal Monovalent Ions | Vary salt (KCl, NaCl) from 0-250 mM. | Rate shows a distinct peak at specific ionic strength. | Include a no-salt condition. |
Detailed Experimental Protocols
Protocol 1: Cofactor Identity and Concentration Optimization
- Objective: Determine the optimal type and concentration of divalent cation.
- Method:
- Prepare 10X stock solutions of MgCl₂, MnCl₂, CaCl₂ (0, 5, 10, 20, 50, 100 mM final in assay).
- In a 96-well plate, mix purified P-loop protein (final 100 nM) in reaction buffer (e.g., 50 mM Tris-HCl, pH 7.5, 100 mM NaCl) with 1 mM ATP/GTP (containing trace [γ-³²P]ATP or using colorimetric assay).
- Initiate reaction by adding the cation stock. Incubate at 30°C.
- Quench at timed intervals (e.g., 0, 2, 5, 10, 20 min) with 5% TCA (for phosphate release) or EDTA (for nucleotide analysis).
- Quantify product (Pi or ADP/GDP). Plot initial velocity (nM product/min) vs. cation concentration.
Protocol 2: Testing for Allosteric Regulation
- Objective: Identify protein or small molecule allosteric regulators.
- Method:
- Purify suspected regulatory protein domain (e.g., a GAP domain for a GTPase).
- Set up standard hydrolysis assay (with optimal Mg²⁺) in the presence of increasing concentrations of the regulator (0, 10, 50, 100, 500 nM).
- Measure initial rates. For single-turnown assays (e.g., mant-GTP hydrolysis), use stopped-flow fluorimetry.
- Plot rate (kobs) vs. [regulator]. A hyperbolic increase indicates allosteric activation. Fit data to the Michaelis-Menten equation to derive an activation constant (Kact).
Pathway Analysis: Integrating Regulation
Diagram Title: Systematic Troubleshooting Workflow for Low Hydrolysis
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Hydrolysis Rate Analysis
| Reagent / Material | Function / Role | Example Product / Note |
|---|---|---|
| Ultrapure MgCl₂ / MnCl₂ | Catalytic cofactor; source of divalent cations. | Sigma-Aldrich Molecular Biology Grade, prepared in Chelex-treated water to remove contaminant cations. |
| ATP/GTP, Regio-specifically Labeled | Hydrolysis substrate for tracking. | [γ-³²P]ATP for sensitive radiometric assays; mant-GTP for fluorescent real-time kinetics. |
| Chelating Resin | Removal of trace divalent cations from buffers. | Chelex 100 Resin; essential for preparing "metal-free" buffers for titration. |
| Phosphate Detection System | Quantifying inorganic phosphate (Pi) release. | Malachite Green Phosphate Assay Kit (colorimetric) or Biomol Green for high-throughput. |
| Stopped-Flow Spectrofluorimeter | Measuring pre-steady-state kinetics of conformational changes & hydrolysis. | Applied Photophysics SX20 or TgK Scientific instrument for rapid mixing (<2 ms). |
| Site-Directed Mutagenesis Kit | Probing conserved motif (P-loop, Walker B) function. | NEB Q5 Site-Directed Mutagenesis Kit for creating alanine substitutions. |
| Gel Filtration Columns | Removing endogenous nucleotides from purified protein. | Cytiva HiPrep 26/10 Desalting column for rapid buffer exchange into nucleotide-free buffer. |
Resolving low hydrolysis rates in NBS domain proteins demands a systematic interrogation of both catalytic cofactors and allosteric circuitry. As detailed in this guide, empirical optimization of the ionic environment, coupled with targeted searches for regulatory partners, directly informs the core thesis on P-loop functional plasticity. These principles are indispensable for accurately reconstituting enzymatic activity in vitro, a foundation for mechanistic understanding and the rational design of allosteric inhibitors in drug development.
Best Practices for Reproducible Functional Characterization of NBS Domains
1. Introduction & Thesis Context The functional characterization of Nucleotide-Binding Site (NBS) domains is a cornerstone of research into innate immunity and regulated cell death. Within the broader thesis on NBS domain conserved motif function, a primary focus is the P-loop (or Walker A motif), responsible for binding and hydrolyzing ATP. Reproducible characterization of its function is critical for elucidating signaling mechanisms in NLR proteins and for validating these domains as targets in drug development for inflammatory diseases and cancer.
2. Core Experimental Workflow for P-loop Functional Analysis A standardized, multi-pronged approach is required to dissect P-loop function. The workflow below integrates biophysical, biochemical, and cellular assays.
Title: Integrated Workflow for NBS P-loop Functional Analysis
3. Detailed Methodologies & Protocols
3.1. Protein Purification (Steps for E. coli system)
- Construct Design: Clone NBS domain (e.g., residues 1-200 of human NLRP3) into a His6-SUMO or GST-tagged vector. Generate P-loop mutant (e.g., K→A in Walker A) via site-directed mutagenesis. Sequence verify.
- Expression: Transform into BL21(DE3) E. coli. Grow culture to OD600 0.6-0.8, induce with 0.5 mM IPTG at 16°C for 16-18 hours.
- Purification: Lyse cells in Lysis Buffer (50 mM Tris pH 8.0, 300 mM NaCl, 10 mM Imidazole, 5% glycerol, 1 mM DTT, protease inhibitors). Clarify lysate. Purify via Ni-NTA affinity chromatography. Elute with 250 mM imidazole.
- Tag Cleavage & Clean-up: Incubate with Ulp1 protease (for SUMO) or Thrombin (for GST) at 4°C overnight. Pass over a second affinity column to remove tag and protease.
- Final Polish: Use size-exclusion chromatography (Superdex 75) in Assay Buffer (20 mM HEPES pH 7.5, 150 mM NaCl, 1 mM TCEP). Concentrate, aliquot, flash-freeze.
3.2. In Vitro Nucleotide Binding Assay (Thermal Shift)
- Principle: P-loop binding stabilizes protein; measure via melting temperature (Tm) shift.
- Protocol: Use a real-time PCR machine. In a 96-well plate, mix 5 µM purified protein with 5X SYPRO Orange dye in Assay Buffer ± 1 mM nucleotide (ATP, ADP, ATPγS). Total volume 25 µL.
- Run: Ramp temperature from 25°C to 95°C at 1°C/min. Monitor fluorescence.
- Analysis: Calculate Tm from the first derivative of the melt curve. ΔTm = Tm(nucleotide) - Tm(apo).
3.3. In Vitro ATPase Activity Assay (Malachite Green)
- Principle: Measure phosphate release from ATP hydrolysis.
- Protocol: In a 96-well plate, incubate 10 µM NBS protein in Assay Buffer + 5 mM MgCl2 with 1 mM ATP at 30°C.
- Quench: At time points (0, 15, 30, 60, 120 min), remove 30 µL reaction and add to 100 µL Malachite Green reagent in a fresh plate.
- Detection: Incubate 20 min, measure A620 nm. Compare to a phosphate standard curve.
- Analysis: Calculate released phosphate (nmol) and determine hydrolysis rate (nmol/min/µg).
3.4. Cellular Signaling Reporter Assay (NF-κB/IRF Luciferase)
- Principle: Test functional consequence of P-loop mutation in full-length protein.
- Protocol: Seed HEK293T cells in 96-well plate. Co-transfect with:
- Expression plasmid for wild-type or P-loop mutant NLR.
- Reporter plasmid (e.g., IFN-β or IL-1β promoter driving firefly luciferase).
- Renilla luciferase control plasmid.
- Stimulation: At 24h post-transfection, stimulate with relevant agonist (e.g., MDP for NOD2, nigericin for NLRP3) or leave unstimulated.
- Lysis & Read: At 48h, lyse cells and measure luminescence using a dual-luciferase kit.
- Analysis: Calculate Firefly/Renilla ratio. Normalize mutant activity to wild-type.
4. Quantitative Data Presentation & Analysis
Table 1: Representative Data from P-loop Functional Characterization
| Assay | Protein Construct | Key Parameter | Wild-type Result (Mean ± SD) | P-loop Mutant Result (Mean ± SD) | Interpretation |
|---|---|---|---|---|---|
| Thermal Shift | NLRP3 NBS (1-200) | ΔTm with ATPγS | +4.2 ± 0.3 °C | +0.5 ± 0.2 °C | Mutant loses nucleotide binding. |
| ATPase Activity | NLRC4 NBS (1-220) | Hydrolysis Rate | 12.5 ± 1.1 nmol/min/µg | 0.8 ± 0.3 nmol/min/µg | Mutant is catalytically inactive. |
| Reporter Assay | Full-length NOD2 | Fold Induction (MDP) | 45.2 ± 5.7 | 1.5 ± 0.4 | P-loop essential for in cellulo signaling. |
| HDX-MS | RIPK2 NBS | % Deuterium Uptake (Loop) | Protected with ADP | No protection with ADP | P-loop mutation disrupts conformational change. |
5. The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents for NBS Domain Characterization
| Reagent / Material | Supplier Examples | Function in Experiments |
|---|---|---|
| His6-SUMO Tag Vector | Addgene, Invitrogen | Enhances soluble expression and enables clean tag removal. |
| Site-Directed Mutagenesis Kit | NEB, Agilent | Creates precise P-loop (e.g., K→A) mutations. |
| Ni-NTA Superflow Resin | Qiagen, Cytiva | Affinity purification of His-tagged recombinant NBS proteins. |
| Superdex 75 Increase | Cytiva | Size-exclusion chromatography for protein polishing and oligomerization state analysis. |
| SYPRO Orange Dye | Thermo Fisher | Fluorescent dye for thermal shift assays to monitor protein unfolding. |
| Malachite Green Kit | Sigma-Aldrich, Abcam | Colorimetric detection of inorganic phosphate for ATPase kinetics. |
| Dual-Luciferase Reporter Kit | Promega | Quantifies pathway-specific transcriptional activation in cell-based assays. |
| Hydrogen-Deuterium Exchange | Waters, Sciex | Mass spectrometry platform for probing nucleotide-induced conformational dynamics. |
6. Signaling Pathway Context Understanding P-loop function requires placing the NBS domain within its native signaling pathway. The diagram below illustrates a generic NLR activation pathway.
Title: NLR Activation Pathway Highlighting Nucleotide Exchange
P-loop in Context: Functional Validation and Cross-Family Comparative Analysis
This whitepaper, framed within a broader thesis on Nucleotide-Binding Site (NBS) domain conserved motifs, details the critical methodologies for validating the in vivo function of the phosphate-binding loop (P-loop). The P-loop, a canonical Walker A motif (GXXXXGK[T/S]), is essential for nucleotide binding and hydrolysis in NBS domains found across proteins like kinases, GTPases, and ATP-binding cassette (ABC) transporters. Dysfunctional P-loops are implicated in numerous diseases, including cancer, neurodegenerative disorders, and immunodeficiency syndromes. This guide provides an in-depth technical framework for two cornerstone approaches: genetic complementation to assess functional restitution and systematic mapping of disease-associated mutations onto the P-loop structure.
The P-loop: Structure, Function, and Disease Context
The P-loop is a topologically conserved loop structure that coordinates the phosphate moiety of ATP or GTP via backbone amide groups and a critical lysine residue. Its proper function is non-negotiable for enzymatic activity and signal transduction.
Table 1: Representative Human Diseases Linked to P-loop Mutations
| Disease | Affected Protein(s) | Common P-loop Mutation(s) | Consequence |
|---|---|---|---|
| Retinitis Pigmentosa | ABCA4 | G863A, GXGKS motif | Impaired retinal ATPase activity, toxic retinoid accumulation. |
| Cystic Fibrosis | CFTR (ABCC7) | G551D, Walker A motif | Disrupted ATP hydrolysis, defective channel gating. |
| Dilated Cardiomyopathy | LMNA | Multiple GXGKS variants | Altered nuclear envelope stability & mechanosignaling. |
| Immunodeficiency | ATM (PI3K) | Multiple Walker A variants | Loss of kinase activity, defective DNA damage response. |
| Cancer (Various) | Multiple Kinases (e.g., BRAF) | G466V, G596R (in P-loop) | Constitutive activation of MAPK pathway. |
Core Methodology I: Genetic Complementation Assay
Genetic complementation is the gold standard for demonstrating that a wild-type (WT) P-loop sequence can rescue a functional defect caused by a mutant P-loop in vivo.
Detailed Experimental Protocol
A. Generation of the Null or P-loop Mutant Background:
- Cell Line/Organism: Select an appropriate model (e.g., yeast knockout strain, mammalian CRISPR-Cas9 knockout cell line, patient-derived mutant cells).
- Validation: Confirm loss of protein expression (via western blot) and associated phenotypic defect (e.g., growth arrest, metabolic deficiency, impaired signaling readout).
B. Complementation Vector Construction:
- Vector Backbone: Use an expression vector with a selectable marker and a promoter suitable for the model system.
- Insert Cloning: Generate three constructs:
- WT: Full-length cDNA with native P-loop sequence.
- P-loop Mutant (MUT): cDNA containing the specific disease-associated mutation (e.g., GXXXXGK[T/S] → AXXXXAK[T/S]).
- Vector Control (VC): Empty vector backbone.
- Optional Control: A catalytically dead mutant outside the P-loop (e.g., DxxxxD/E Walker B mutant).
C. Transfection/Transformation & Selection:
- Introduce constructs into the null/mutant background using standardized methods (electroporation, lipid transfection, lithium acetate transformation).
- Apply appropriate selection (antibiotics, auxotrophic selection) for 5-7 days to establish stable polyclonal populations or isolate individual clones.
D. Functional Phenotyping (Quantitative Readouts):
- Growth/Rescue Assay: Plate equal numbers of cells and measure colony formation, optical density (OD600), or confluence over 72-96 hours.
- Biochemical Activity: Perform in vitro kinase/GTPase/ATPase assays on immunoprecipitated proteins from complemented cells.
- Pathway Activity: Quantify downstream phosphorylation (e.g., p-ERK for MAPK pathway) via phospho-specific western blot or ELISA.
- Cellular Phenotype: For disease-specific models, assay restored function (e.g., iodide efflux for CFTR, DNA repair foci for ATM).
E. Data Analysis:
- Normalize all quantitative data to the vector control (set as 0% rescue) and the wild-type complement (set as 100% rescue).
- Statistical significance is typically assessed via one-way ANOVA with post-hoc tests (n≥3).
Diagram 1: Genetic complementation experimental workflow.
Core Methodology II: Disease Mutation Mapping & Analysis
Systematic mapping of patient-derived mutations onto the P-loop structure provides mechanistic insights and stratifies variants of unknown significance (VUS).
Detailed Experimental Protocol
A. Data Curation:
- Source Variants: Extract missense mutations within the P-loop consensus sequence from public databases (ClinVar, gnomAD, cBioPortal) and literature.
- Filtering: Categorize mutations as: (i) Pathogenic/Likely Pathogenic, (ii) Benign/Likely Benign, (iii) VUS.
B. Structural Mapping & In Silico Analysis:
- Template Acquisition: Obtain a high-resolution crystal structure of the target protein's NBS domain (preferably with bound nucleotide) from the PDB (e.g., 1ATP).
- Visualization & Analysis: Use software (PyMOL, ChimeraX) to:
- Map mutation positions onto the 3D structure.
- Measure distances between mutated residues and the nucleotide (α/β-phosphate) or catalytic Mg²⁺ ion.
- Analyze potential disruptions in hydrogen bonding or salt bridges.
- Perform computational stability predictions (ΔΔG) using tools like FoldX or Rosetta.
C. In Vitro Biochemical Validation of Mapped Mutants:
- Protein Purification: Express and purify recombinant WT and selected mutant proteins (e.g., His-tagged NBS domain) from E. coli or insect cells.
- Nucleotide Binding Affinity:
- Method: Isothermal Titration Calorimetry (ITC) or Fluorescence Anisotropy.
- Protocol (Anisotropy): Label nucleotide (e.g., Mant-ATP). Incubate with increasing protein concentrations (0-100 µM) in assay buffer. Measure anisotropy. Fit data to a quadratic binding equation to derive Kd.
- Catalytic Activity:
- Method: Malachite Green Phosphate Release or coupled enzymatic assay.
- Protocol (Malachite Green): Incubate protein (100 nM) with ATP (1 mM) in reaction buffer at 30°C. At intervals, stop reaction with malachite green reagent. Measure A620. Compare initial velocities (V0).
Table 2: Exemplary Mutation Mapping Data for a Hypothetical Kinase P-loop
| Mutation | ClinVar Classification | Distance to ATP γ-P (Å) | Predicted ΔΔG (kcal/mol) | Measured Kd for ATP (µM) | Catalytic Rate (kcat, s⁻¹) |
|---|---|---|---|---|---|
| WT | - | 4.2 | 0.0 | 15.2 ± 1.5 | 25.0 ± 2.1 |
| G12S | Pathogenic | 6.8 | +2.3 | >200 | 0.5 ± 0.1 |
| K15E | Pathogenic | 12.5 (Salt bridge loss) | +4.1 | N.B.* | N.D. |
| G12A | VUS | 5.1 | +0.8 | 45.3 ± 4.2 | 8.7 ± 1.2 |
| T17A | Benign | 7.0 | -0.2 | 18.5 ± 2.0 | 22.1 ± 1.8 |
N.B.: No detectable binding. *N.D.: Not detectable.
Integrating Complementation & Mapping: A Case Study Framework
The combined approach conclusively links genotype, molecular phenotype, and cellular function.
Diagram 2: Integrated workflow for P-loop mutation analysis.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents and Materials for P-loop Functional Studies
| Item | Function & Application | Example/Details |
|---|---|---|
| CRISPR/Cas9 Knockout Kit | Generation of isogenic P-loop null cell lines for complementation. | Lentiviral sgRNA vectors targeting the Walker A motif; includes puromycin selection. |
| Site-Directed Mutagenesis Kit | Rapid introduction of P-loop mutations into cDNA constructs. | High-fidelity polymerase mix, DpnI enzyme, optimized for G-C rich motifs. |
| Mammalian Expression Vector | Delivery of WT/mutant constructs for complementation. | CMV or EF1α promoter, C-terminal FLAG/HA tag, hygromycin resistance. |
| Mant-ATP (2'/3'-O-(N-Methylanthraniloyl)) | Fluorescent ATP analog for direct binding affinity measurements. | Used in fluorescence anisotropy/FRET-based nucleotide binding assays. |
| Malachite Green Phosphate Assay Kit | Colorimetric detection of inorganic phosphate released from ATPase/GTPase reactions. | Sensitive to 0.1 nmol phosphate; ideal for kinetic analysis of purified proteins. |
| Anti-Phospho-Substrate Antibodies | Readout of downstream pathway activity rescue in complementation assays. | e.g., Phospho-(Ser/Thr) PKA substrate Ab for kinase P-loop function. |
| Recombinant Protein Purification System | Production of pure WT and mutant NBS domains for in vitro biochemistry. | HisTrap column kits for immobilized metal affinity chromatography (IMAC). |
| Structural Visualization Software | Mapping mutations and analyzing interactions in 3D. | PyMOL or UCSF ChimeraX licenses for academic/industrial use. |
Within the broader thesis on NBS (Nucleotide-Binding Site) domain conserved motif function, the Walker A motif, or P-loop, serves as a fundamental functional unit for nucleotide binding and hydrolysis across diverse protein superfamilies. This whitepaper provides a comparative, in-depth analysis of P-loop mechanics in two distinct systems: ATP-Binding Cassette (ABC) transporters, which are integral membrane proteins involved in substrate translocation, and Nod-like Receptor (NLR) immune receptors, which are cytosolic sensors of pathogen invasion and cellular stress. Despite sharing a conserved phosphate-binding loop (G-X-X-X-X-G-K-T/S), the structural context, allosteric regulation, and functional outcomes of nucleotide binding differ profoundly. This guide details the molecular mechanisms, experimental approaches, and quantitative data defining P-loop function in these families, providing a resource for researchers in structural biology, immunology, and drug discovery.
Core Molecular Mechanics and Structural Context
ABC Transporters
In ABC transporters, the P-loop is part of a canonical nucleotide-binding domain (NBD). ATP binding occurs at the interface of two NBDs, sandwiching the nucleotide between the Walker A motif of one NBD and the Signature (LSGGQ) motif of the opposing NBD. ATP binding induces NBD dimerization, a key step that powers conformational changes in the transmembrane domains for substrate translocation. Hydrolysis, facilitated by a catalytic glutamate from the Walker B motif and a Mg²⁺ ion coordinated by the P-loop, resets the cycle.
NLR Immune Receptors
In NLRs (e.g., NLRP3, NOD2), the P-loop resides within a central NACHT domain (NAIP, CIITA, HET-E, and TP1). Nucleotide binding (typically ATP/dATP) is primarily a regulatory switch controlling the transition from an auto-inhibited monomer to an active, oligomeric inflammasome or signalosome. The P-loop mediates nucleotide binding, but hydrolysis often serves a down-regulatory or disassembly function. Mutations in the NACHT domain P-loop are linked to autoinflammatory diseases.
Table 1: Quantitative Comparison of P-loop Characteristics
| Parameter | ABC Transporters | NLR Immune Receptors |
|---|---|---|
| Primary Nucleotide | ATP | ATP/dATP |
| Typical Kd for Nucleotide | 1-100 µM | 10-500 µM (highly variable) |
| Hydrolysis Rate (kcat) | 1-100 s⁻¹ | 0.1-5 s⁻¹ (often slower, regulatory) |
| Key Coordinating Residues | Walker A Lys, Mg²⁺, Walker B Glu | Walker A Lys, Mg²⁺, Sensor-1 Arg/His |
| Functional Outcome | Mechanical work (translocation) | Oligomerization & Signal Activation |
| Disease Link | Cystic Fibrosis (CFTR), Drug Resistance | CAPS (NLRP3), Blau Syndrome (NOD2) |
Key Experimental Protocols
Nucleotide Binding Affinity Assay (Fluorescence Polarization)
Purpose: To determine dissociation constant (Kd) for ATP binding to purified NBD/NACHT domains. Protocol:
- Protein Purification: Express and purify recombinant protein containing the NBS domain (e.g., NBD of ABCB1 or NACHT domain of NLRP3) with an N-terminal His-tag.
- Tracer Preparation: Use fluorescent ATP analogue (e.g., BODIPY-FL-ATP, TNP-ATP).
- Assay Setup: In a 96-well plate, hold fluorescent tracer concentration constant (e.g., 10 nM) and titrate purified protein across a range (e.g., 0 nM to 200 µM) in binding buffer (20 mM HEPES pH 7.5, 150 mM NaCl, 5 mM MgCl₂).
- Incubation & Measurement: Incubate 30 min at 4°C. Measure fluorescence polarization (mP units) using a plate reader.
- Analysis: Fit data to a one-site binding model:
mP = mP_min + (mP_max - mP_min) * [P] / (Kd + [P]), where [P] is protein concentration.
ATPase Activity Assay (Coupled Enzymatic)
Purpose: To measure steady-state ATP hydrolysis kinetics (kcat, KM). Protocol:
- Reaction Mix: Prepare a master mix containing final concentrations of 50 mM Tris-HCl pH 7.5, 50 mM KCl, 5 mM MgCl₂, 1 mM DTT, 2 mM phosphoenolpyruvate (PEP), 0.2 mM NADH, 10 U/mL pyruvate kinase, 10 U/mL lactate dehydrogenase.
- Initiation: Add purified protein (10-100 nM) and varying ATP concentrations (0.05 to 5 mM) to the master mix.
- Monitoring: Read absorbance at 340 nm continuously for 30-60 min at 37°C. The oxidation of NADH to NAD⁺ causes a decrease in A340 proportional to ATP hydrolyzed.
- Calculation: Convert ΔA340/min to µmol ATP/min using NADH's extinction coefficient (6220 M⁻¹cm⁻¹). Plot rate vs. [ATP] and fit to the Michaelis-Menten equation.
Inflammasome Activation Assay (Cellular)
Purpose: To assess functional impact of P-loop mutations in NLRs. Protocol:
- Cell Culture: Seed THP-1 cells (human monocytic line) or primary macrophages in 96-well plates. Differentiate with PMA (100 nM, 24h).
- Transfection/Mutation: Transfect with plasmids expressing wild-type or P-loop mutant NLRP3 (e.g., K-to-A in Walker A). Alternatively, use CRISPR-edited cell lines.
- Stimulation: Prime cells with LPS (1 µg/mL, 4h) then activate with nigericin (10 µM, ATP (5 mM), or other NLRP3 agonists.
- Readout: Measure IL-1β in supernatant via ELISA 1h post-activation. Assess cell death via LDH release assay. For direct visualization, perform immunoblotting for cleaved caspase-1.
Visualizations
Title: ABC Transporter ATPase Cycle
Title: NLRP3 Inflammasome Activation Pathway
Title: P-loop Functional Study Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for P-loop Research
| Item | Function & Application | Example Product/Catalog |
|---|---|---|
| Fluorescent ATP Analog | Tracer for binding affinity measurements via Fluorescence Polarization (FP). | BODIPY FL ATP-γ-S (ThermoFisher, B32700); TNP-ATP (Jena Bioscience, NU-901) |
| Coupled Enzyme ATPase Kit | For continuous, spectrophotometric measurement of ATP hydrolysis kinetics. | ATPase/GTPase Activity Assay Kit (Sigma, MAK113) |
| Anti-Phospho-Antibodies | Detect phosphorylated intermediates (e.g., P-loop adjacent phospho-regulation). | Anti-Phospho-(Ser/Thr) Antibodies (Cell Signaling Tech) |
| Non-Hydrolyzable ATP Analog | To trap nucleotide-bound states for structural studies or inhibit hydrolysis. | ATP-γ-S (Sigma, A1388); AMP-PNP (Sigma, A2647) |
| Site-Directed Mutagenesis Kit | Engineer point mutations in Walker A (P-loop) sequences (e.g., K→A, T→A). | Q5 Site-Directed Mutagenesis Kit (NEB, E0554S) |
| NLR Inflammasome Activation Kits | Cellular assays for NLR function downstream of P-loop. | NLRP3 Inflammasome Assay Kit (InvivoGen, rep- nlr p3); IL-1β ELISA Kit (R&D Systems, DLB50) |
| Membrane Protein Detergent | Solubilize and stabilize ABC transporters for in vitro studies. | n-Dodecyl-β-D-Maltopyranoside (DDM, GoldBio, DDM25) |
| Cryo-EM Grids | For high-resolution structural analysis of nucleotide-bound states. | Quantifoil R1.2/1.3 Au 300 mesh grids (Electron Microscopy Sciences) |
This whitepaper provides a technical guide to the core structural motifs of the Nucleotide-Binding Site (NBS) domain, framing their integrated function within the context of broader research into P-loop mechanism and evolution. The NBS is a conserved module critical for ATP/GTP hydrolysis in numerous protein families, including ABC transporters, kinases, GTPases, and NLR immune receptors. Its function emerges not from isolated motifs, but from the precise spatial and energetic coordination between the phosphate-binding loop (P-loop/Walker A), the Walker B motif, and additional "Signature" motifs. Understanding this integrated machinery is paramount for researchers and drug development professionals targeting these domains in diseases ranging from cancer to autoimmunity.
Core Motifs: Definition and Canonical Sequence
| Motif Name | Canonical Sequence (Amino Acids) | Primary Structural Role | Key Functional Atoms/Groups |
|---|---|---|---|
| P-loop (Walker A) | G-X-X-X-X-G-K-T/S | Binds α- and β-phosphates of NTP | Backbone amides (N), Lys/Ser/Thr side chain |
| Walker B | h-h-h-h-D/E (h=hydrophobic) | Coordinates Mg²⁺ via acidic residue; promotes hydrolysis | Asp/Glu (Mg²⁺ coordination), hydrophobic stack |
| Signature Motif | Varied (e.g., LSGGQ in ABC exporters, C-loop in Kinases) | Contributes to nucleotide specificity & inter-domain communication | Diverse, often provides sandwiching aromatic or specific H-bonds |
Integrated Mechanistic Function
The hydrolysis cycle is a concerted process:
- Nucleotide Binding: The P-loop's flexible loop conformation closes upon NTP binding, positioning the β-phosphate. The invariant Lys/Ser stabilizes the negatively charged transition state.
- Mg²⁺ and Water Activation: The Walker B aspartate/glutamate chelates the essential Mg²⁺ ion, which in turn positions and polarizes the nucleophilic water molecule.
- Catalytic Transition State Stabilization: The P-loop lysine (or serine/threonine) and the Mg²⁺ ion work synergistically to neutralize negative charge buildup during hydrolysis. The Signature motif often forms a "lid" or provides additional contacts that stabilize the closed, active conformation.
- Product Release & Conformational Change: Following γ-phosphate cleavage, the motifs collectively facilitate ADP/Pi release, often coupled to large-scale domain movements (e.g., in ABC transporters or NLR activation).
Diagram Title: Integrated NBS Motif Catalytic Cycle
Table 1: Energetic and Kinetic Contributions of NBS Motifs (Representative Data)
| Protein System | Mutation (Motif) | ΔKd(NTP) (Fold Increase) | kcat Reduction (Fold) | Experimental Method | Reference (Type) |
|---|---|---|---|---|---|
| p21 Ras (GTPase) | K16A (P-loop Lys) | ~10³ | >10⁵ | Stopped-flow, ITC | Wittinghofer, 1997 |
| ABC Transporter BtuD | S43L (P-loop Ser) | 5 | 100 | ATPase assay, Crystallography | Locher, 2002 |
| NLR Protein APAF-1 | D397A (Walker B) | Mild | ~10³ (No oligomerization) | SEC-MALS, ATPase assay | Bax, 2016 |
| Kinase PKA | D166N (Walker B) | N/A | ~10⁴ | Radioactive ATPase assay | Taylor, 1992 |
| Bacterial RecA | Δ Signature (C-loop) | >10² | Not determined | Fluorescence Polarization | Cox, 2008 |
Table 2: Conserved Structural Parameters from Crystallographic Surveys
| Parameter | P-loop Interaction | Walker B Interaction | Signature Motif Role |
|---|---|---|---|
| Distance to β-P (Å) | 1.6 - 2.0 (Backbone N) | 4.0 - 4.5 (via Mg²⁺) | Variable |
| Distance to γ-P/Water (Å) | 3.0 - 3.5 (Lys NZ / Thr OG1) | 2.0 - 2.2 (Mg²⁺ to Water) | Often >5 Å (allosteric) |
| Bond Angle (Catalytic) | O1P-β-P-O2P ~120° | Mg²⁺-Owater-Hwater ~180° | N/A |
| Conformational Change | Loop closure >5 Å | Helix/strand shift ~2 Å | Domain rotation up to 30° |
Key Experimental Protocols
5.1. Site-Directed Mutagenesis & Recombinant Protein Purification for Motif Analysis
- Objective: Generate specific mutations (e.g., K→A in P-loop, D→N in Walker B) in the NBS domain.
- Protocol:
- Design primers with the desired mutation for PCR-based mutagenesis (e.g., QuikChange).
- Transform plasmid into competent E. coli cloning strain, sequence-verify the mutation.
- Express recombinant protein in a suitable system (e.g., E. coli BL21(DE3), insect cells).
- Purify via affinity chromatography (Ni-NTA for His-tag, glutathione resin for GST-tag) followed by size-exclusion chromatography (SEC) for monodispersity.
- Confirm folding via circular dichroism (CD) spectroscopy.
5.2. Isothermal Titration Calorimetry (ITC) for Nucleotide Binding Affinity
- Objective: Quantify the dissociation constant (Kd), stoichiometry (n), and thermodynamics (ΔH, ΔS) of NTP binding to wild-type vs. mutant NBS domains.
- Protocol:
- Dialyze purified protein and nucleotide (ATP/GTP) into identical, degassed buffer.
- Load the protein solution (typically 50-100 μM) into the sample cell.
- Fill the syringe with nucleotide solution (10x concentrated relative to protein).
- Perform automated injections at constant temperature (e.g., 25°C). A control (nucleotide into buffer) is subtracted.
- Fit the integrated heat data to a single-site binding model to extract Kd and ΔH.
5.3. Malachite Green Phosphate Release Assay for Hydrolysis Kinetics
- Objective: Measure steady-state ATPase/GTPase kinetics (kcat, KM).
- Protocol:
- Prepare reaction buffer containing protein and varying concentrations of NTP (Mg²⁺ present).
- Incubate at defined temperature, taking aliquots at time intervals.
- Stop the reaction by adding the Malachite Green reagent (ammonium molybdate in sulfuric acid with malachite green).
- Measure absorbance at 620-660 nm after color development (15-30 min).
- Compare to a standard curve of inorganic phosphate (Pi) to calculate released Pi over time.
- Plot initial velocity vs. [NTP] and fit to the Michaelis-Menten equation.
5.4. Crystallography for Structural Determination of Intermediate States
- Objective: Obtain high-resolution structures of NBS domains with bound substrates (ATP/GTP), transition-state analogs (ADP·VO₄, ADP·AlF₃), or products (ADP).
- Protocol:
- Co-crystallize protein with nucleotide and Mg²⁺ (or analog).
- Flash-cool crystal in liquid N₂ with cryoprotectant.
- Collect X-ray diffraction data at a synchrotron source.
- Solve structure by molecular replacement using a homologous NBS domain.
- Refine model; analyze electron density for ligand placement, bond distances, and conformational changes in motifs.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for NBS Motif Functional Analysis
| Reagent / Material | Function / Application | Key Considerations |
|---|---|---|
| Transition-State Analogs (ADP·VO₄, ADP·AlF₃, GDP·AlF₄⁻) | Trap the catalytic site in a hydrolytic transition state for crystallography or binding studies. | AlF₃ analogs mimic pentavalent γ-phosphate; Vanadate is a planar PO₃⁻ analog. Prepare fresh. |
| Non-hydrolyzable NTP Analogs (ATPγS, AMP-PNP, GMP-PNP) | Study nucleotide binding without hydrolysis, isolating the first step of the cycle. | Binding affinity may differ from natural NTPs. Check for residual activity. |
| Malachite Green Phosphate Assay Kit | Colorimetric detection of inorganic phosphate (Pi) released during hydrolysis. | Sensitive to free Pi contamination. Use high-purity water and buffers. |
| Size-Exclusion Chromatography (SEC) Columns (e.g., Superdex 200 Increase) | Assess protein oligomerization state, which is often coupled to NBS status (e.g., NLR activation). | Calibrate with standard proteins. Run in assay-relevant buffer. |
| Fluorescent NTP Analogs (e.g., Mant-ATP, TNP-ATP) | Monitor real-time binding and conformational changes via fluorescence polarization or FRET. | The fluorophore moiety can alter binding kinetics; always compare to native NTP. |
| ITC-Compatible High-Purity Buffers | Ensure accurate thermodynamic measurements by minimizing heat of dilution. | Use same buffer batch for protein and ligand dialysis. Avoid high detergent concentrations. |
This whitepaper serves as a technical guide to the structural and functional dynamics of G-domain loops in GTPases, framed within the foundational research on the nucleotide-binding site (NBS). The P-loop (Walker A motif), a critical component of the NBS, is a conserved glycine-rich loop that coordinates the phosphate moieties of bound nucleotides. Understanding its function in GTPases—proteins that act as molecular switches in essential cellular pathways—provides profound insights into mechanistic biology and therapeutic intervention. This analysis draws parallels and highlights divergences between distinct GTPase families, focusing on the functional interplay between the P-loop and other conserved G-domain motifs (Switch I, Switch II, and G motifs).
The G-Domain: Core Loops and Motifs
The GTPase domain (G-domain) is defined by a set of conserved sequence motifs that form specific loops and structural elements essential for nucleotide binding, hydrolysis, and conformational switching. These loops are not isolated; their functions are deeply integrated.
- P-loop (G1/GKS motif): Binds the α- and β-phosphates of GTP/GDP. Its main chain amides are critical for stabilizing the transition state during hydrolysis.
- Switch I (G2 motif): Typically contains a conserved Thr/Ser residue that coordinates the Mg²⁺ ion and the γ-phosphate. It is a key sensor of nucleotide state.
- Switch II (G3 motif): Contains a catalytically essential glutamine (Gln⁶¹ in Ras) that positions the hydrolytic water molecule. Its conformation is nucleotide-dependent.
- G4 motif (NKxD): Specifics guanine base recognition via hydrogen bonding.
- G5 motif (SAK/L): Further stabilizes the guanine base.
Table 1: Conserved Motifs and Primary Functions in Canonical GTPases
| Motif Name | Consensus Sequence | Primary Structural Role | Key Interactions |
|---|---|---|---|
| P-loop (G1) | GxxxxGK[S/T] | Phosphate binding | α-/β-phosphate, Mg²⁺ |
| Switch I (G2) | xT/Sx | Mg²⁺/γ-phosphate sensor | Mg²⁺, γ-phosphate, effectors |
| Switch II (G3) | DxxGQ | Hydrolysis catalysis | Gln stabilizes H₂O, Gly allows conformational flexibility |
| G4 (NKxD) | N/TKxD | Guanine specificity | H-bonds to guanine base |
| G5 (SAK/L) | [S/A]xx | Guanine base stabilization | Hydrophobic/stacking interactions |
Parallels: Conserved Mechanistic Principles
The core hydrolytic mechanism is conserved across many GTPase families (e.g., Ras, Ran, Gα). The P-loop universally provides a rigid scaffold for anchoring the nucleotide. The critical step involves the Gln from Switch II and an exogenous catalytic residue (often an "Arginine Finger" from a GTPase-Activating Protein, GAP). This creates a convergent catalytic apparatus.
Table 2: Quantitative Parameters of GTP Hydrolysis for Selected GTPases
| GTPase | Intrinsic kcat (min⁻¹) | GAP-Stimulated kcat (min⁻¹) | Fold Stimulation | Key Catalytic Residue (External Source) |
|---|---|---|---|---|
| Ras (H-Ras) | ~0.02 | ~10,000 (p120GAP) | ~5 x 10⁵ | Arg⁷⁸⁹ (GAP) |
| RhoA | ~0.03 | ~5,000 (p50RhoGAP) | ~1.7 x 10⁵ | Arg²⁸⁵ (GAP) |
| Gαi | ~0.05 | ~5 (RGS) | ~100 | N/A (RGS stabilizes Switch I/II) |
| Ran | <0.01 | >10,000 (RanGAP) | >10⁶ | Arg⁶⁰⁻⁷⁷ (RanGAP) |
Title: GTPase Activation/Deactivation Cycle
Divergences: Specialization of Loop Function
Divergences arise from specialized cellular roles. For instance, in elongation factor Tu (EF-Tu), the P-loop and Switch I undergo dramatic conformational changes upon GTP hydrolysis, releasing aminoacyl-tRNA. In dynamin-like GTPases, the G-domain loops mediate oligomerization, where the P-loop of one monomer interacts with the Switch II of another, coupling GTP hydrolysis to mechanical constriction. In Gα subunits, the P-loop is a key site for ADP-ribosylation by bacterial toxins (e.g., Pertussis toxin), altering function. These examples illustrate how the core NBS architecture is adapted for diverse outputs.
Experimental Protocols for G-domain Loop Analysis
Protocol 1: Steady-State GTPase Activity Assay (Radioactive)
- Objective: Measure intrinsic and GAP-stimulated hydrolysis rates.
- Materials: Purified GTPase, [γ-³²P]GTP, GAP protein, Charcoal slurry, Reaction buffer (50 mM Tris-HCl pH 7.5, 50 mM NaCl, 5 mM MgCl₂).
- Method:
- Prepare reaction mixtures with 1 µM GTPase, 100 µM [γ-³²P]GTP (± GAP).
- Incubate at 30°C. Aliquot at time points (e.g., 0, 2, 5, 10, 20 min).
- Stop reaction with 5% charcoal slurry in 50 mM NaH₂PO₄ (pH 3.0) on ice. Charcoal binds nucleotide, free ³²Pi remains in supernatant.
- Centrifuge, quantify radioactivity in supernatant via scintillation counting.
- Plot ³²Pi released vs. time. The slope gives the hydrolysis rate.
Protocol 2: Site-Directed Mutagenesis of P-loop/Switch Residues
- Objective: Probe the functional role of specific loop residues.
- Method:
- Design primers to mutate key residues (e.g., G12 in P-loop, Q61 in Switch II of Ras).
- Perform PCR-based mutagenesis (e.g., QuikChange) on GTPase expression plasmid.
- Sequence the plasmid to confirm the mutation.
- Express and purify mutant protein.
- Characterize via GTPase assay (Protocol 1), nucleotide binding (filter binding), and structural analysis (X-ray crystallography).
Protocol 3: X-ray Crystallography of G-domain Complexes
- Objective: Determine atomic structures of GTPase states.
- Method:
- Purify GTPase protein at high concentration (>10 mg/mL).
- Prepare complexes: load with non-hydrolyzable GTP analogs (GMPPNP, GMPPCP) or GDP.
- Co-crystallize with binding partners (GAPs, GEFs, effectors).
- Screen crystallization conditions using commercial sparse-matrix screens.
- Collect diffraction data at a synchrotron source, solve structure via molecular replacement.
- Analyze electron density for P-loop, Switch I, and Switch II conformations.
Title: G-domain Functional Analysis Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for G-domain Studies
| Reagent / Material | Function / Application | Example / Note |
|---|---|---|
| Non-hydrolyzable GTP Analogs | Trap GTPase in "active" conformation for structural/ binding studies. | GMPPNP, GMPPCP. GTPγS is slowly hydrolyzable. |
| Fluorescent Nucleotides | Real-time monitoring of nucleotide binding and dissociation kinetics. | Mant-GTP/GDP, BODIPY-GTP. Fluorescence change upon binding. |
| GTPase Expression Systems | High-yield production of recombinant wild-type and mutant proteins. | E. coli (e.g., BL21), Baculovirus/Sf9 for mammalian GTPases. |
| GST/GST-Tag Vectors | Facilitate purification and pull-down assays for interaction studies. | pGEX series; used to express GTPase or GAP/GEF partners. |
| Anti-GTPase Antibodies | Detection, immunoprecipitation, and cellular localization. | Isoform-specific antibodies critical for Ras, Rho, Rab families. |
| GEF/GAP Protein Assays | Measure nucleotide exchange or hydrolysis stimulation in vitro. | Use fluorescent nucleotides or radioactive assays with purified components. |
| Crystallography Screens | Identify initial conditions for protein crystal growth. | Commercial screens (e.g., Hampton Research, Molecular Dimensions). |
| GTPase Inhibitors | Tool compounds for functional disruption in cells. | ML141 (Cdc42), CID1067700 (Rab7), Rhosin (RhoA). |
The study of G-domain loops, anchored by the P-loop of the NBS, reveals a elegant paradigm of conserved core mechanics coupled to specialized functional adaptations. The P-loop is more than a passive anchor; its precise geometry influences catalytic efficiency and is a site for pathogenic modification. Divergences in Switch region dynamics create family-specific vulnerabilities. Modern drug discovery, particularly against "undruggable" targets like Ras, exploits these lessons. Strategies include: 1) targeting the nucleotide-binding pocket in inactive states, 2) disrupting protein-protein interfaces critical for GEF/GAP action, and 3) exploiting synthetic lethality in downstream pathways. A deep understanding of loop parallels and divergences thus provides a critical blueprint for rational therapeutic design in oncology, neurology, and infectious disease.
The nucleotide-binding site (NBS) domain, characterized by conserved phosphate-binding loop (P-loop) motifs, is a critical functional module in numerous enzymes, including kinases, GTPases, and ATPases. This structural element facilitates nucleotide triphosphate binding and hydrolysis, driving essential cellular processes. Disruption of P-loop function through targeted inhibition has emerged as a powerful therapeutic strategy in oncology and beyond. This whitepaper, framed within broader research on NBS domain motifs, provides an in-depth technical analysis of validated P-loop-targeting drugs, supported by experimental protocols, quantitative data, and essential research tools.
Validated Drug Case Studies
The following table summarizes key quantitative data for three clinically successful P-loop-targeting drugs.
Table 1: Quantitative Profile of Selected P-loop-Targeting Drugs
| Drug Name (Target) | Indication(s) | IC50 / Kd Value | Key P-loop Interaction | Clinical Status |
|---|---|---|---|---|
| Imatinib (Bcr-Abl) | CML, GIST | 200-600 nM (Cellular) | Hydrogen bonds with backbone amides of A380 (P-loop) & E286 | Approved (2001) |
| Sotorasib (KRASG12C) | NSCLC with KRAS G12C mutation | 21.8 nM (Kd for KRASG12C-GDP) | Covalent binding to Cys12 adjacent to P-loop (G10VC12) | Approved (2021) |
| Gefitinib (EGFR) | NSCLC with EGFR mutations | 2-33 nM (Mutant EGFR) | Forms hydrogen bonds with hinge region & P-loop residues (M769) | Approved (2003) |
Experimental Protocols for Validation
3.1. Protocol: Isothermal Titration Calorimetry (ITC) for Binding Affinity Measurement Objective: Determine the binding constant (Kd), enthalpy (ΔH), and stoichiometry (n) of a drug interacting with its P-loop-containing target protein. Materials: VP-ITC or similar instrument, purified target protein (≥95% purity), drug compound, matched dialysis buffer. Procedure:
- Dialyze protein and drug compound extensively against the same degassed assay buffer (e.g., 20 mM HEPES, pH 7.5, 150 mM NaCl).
- Load the cell with protein solution (10-50 μM). Load the syringe with drug solution (10-20 times the protein concentration).
- Set experimental parameters: 25°C, reference power of 10-15 μcal/sec, stirring at 750 rpm.
- Perform titration: Inject 2-10 μL aliquots of drug solution into the protein cell at 180-240 second intervals.
- Analyze data using instrument-specific software (e.g., MicroCal PEAQ-ITC). Fit the corrected heat vs. molar ratio curve to a single-site binding model to derive Kd, ΔH, and n.
3.2. Protocol: Cellular Thermal Shift Assay (CETSA) for Target Engagement Objective: Validate drug binding to the intended P-loop target in a cellular context by measuring thermal stabilization. Materials: Cultured cell line, drug compound, DMSO, protease/phosphatase inhibitors, thermal cycler, Western blot apparatus. Procedure:
- Treat cell aliquots (1-2 million cells) with drug or DMSO control for a predetermined time (e.g., 2 hours).
- Aliquot treated cell suspensions into PCR tubes. Heat each aliquot at distinct temperatures (e.g., 37°C to 67°C, in 3°C increments) for 3 minutes in a thermal cycler.
- Snap-freeze tubes in liquid nitrogen. Thaw and lyse cells using freeze-thaw cycles or detergent-based lysis buffer with inhibitors.
- Centrifuge lysates at high speed (20,000 x g) to separate soluble protein. Analyze the supernatant by SDS-PAGE and Western blot for the target protein.
- Quantify band intensity. Plot soluble protein fraction vs. temperature. A rightward shift in the melting curve for drug-treated samples indicates target engagement and stabilization.
Pathway and Workflow Visualizations
Title: Signaling Pathway Disruption by P-loop Inhibitors
Title: P-loop Drug Development Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for P-loop-Targeting Drug Research
| Reagent / Material | Function & Application | Key Provider Examples |
|---|---|---|
| Recombinant P-loop Protein (Kinase/GTPase) | High-purity target for in vitro binding (ITC, SPR) and enzymatic assays. | SignalChem, BPS Bioscience, Thermo Fisher |
| TR-FRET Kinase Assay Kits | Homogeneous, high-throughput screening for kinase activity inhibition. | Cisbio, Thermo Fisher |
| Active RAS Pull-Down Kit | Detects levels of active, GTP-bound RAS in cell lysates to assess inhibitor efficacy. | Thermo Fisher, Cytoskeleton, Inc. |
| Phospho-Specific Antibodies (e.g., p-CRKL, p-ERK) | Detect pathway inhibition downstream of the P-loop target via Western blot or ELISA. | Cell Signaling Technology, Abcam |
| KRAS G12C Mutant Cell Line (e.g., NCI-H358) | Cellular model for validating mutant-specific KRAS inhibitors like sotorasib. | ATCC |
| CETSA Kit (Commercial) | Standardized reagents and protocols for cellular target engagement studies. | Thermo Fisher |
| Cryo-EM Grids (Quantifoil) | For high-resolution structural determination of drug-target complexes. | Electron Microscopy Sciences |
Conclusion
The conserved P-loop motif is not merely a structural feature but the dynamic, functional heart of the NBS domain, essential for nucleotide binding and energy transduction across a vast array of proteins. Through foundational understanding, robust methodological analysis, careful troubleshooting, and comparative validation, its pivotal role in cellular machinery and disease pathogenesis becomes unequivocally clear. This synthesis underscores the P-loop as a prime, validated target for precision drug development. Future directions must leverage high-resolution dynamics, AI-driven prediction of allosteric networks, and the development of next-generation, selective inhibitors that modulate P-loop function to treat cancer, genetic disorders, and infectious diseases, moving from mechanistic insight to clinical impact.